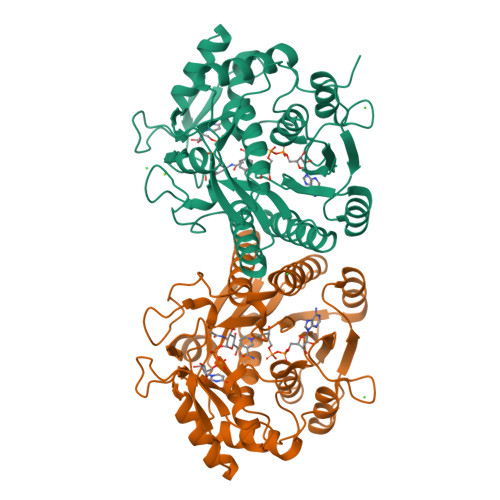
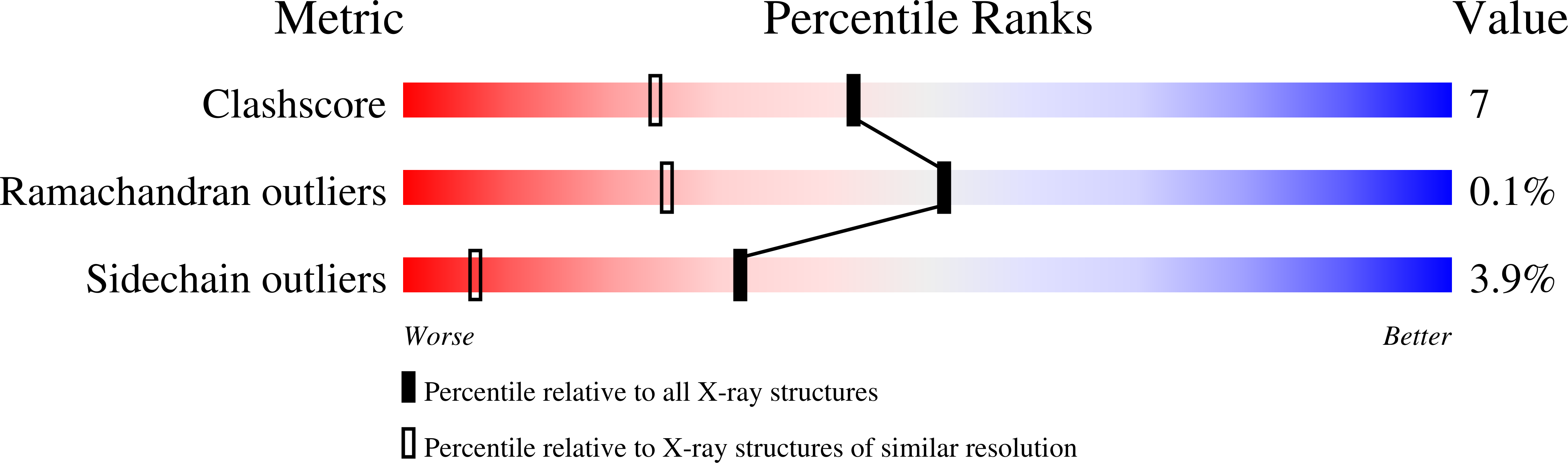Molecular basis for severe epimerase deficiency galactosemia. X-ray structure of the human V94m-substituted UDP-galactose 4-epimerase.
Thoden, J.B., Wohlers, T.M., Fridovich-Keil, J.L., Holden, H.M.(2001) J Biological Chem 276: 20617-20623
- PubMed: 11279193
- DOI: https://doi.org/10.1074/jbc.M101304200
- Primary Citation of Related Structures:
1I3K, 1I3L, 1I3M, 1I3N - PubMed Abstract:
Galactosemia is an inherited disorder characterized by an inability to metabolize galactose. Although classical galactosemia results from impairment of the second enzyme of the Leloir pathway, namely galactose-1-phosphate uridylyltransferase, alternate forms of the disorder can occur due to either galactokinase or UDP-galactose 4-epimerase deficiencies. One of the more severe cases of epimerase deficiency galactosemia arises from an amino acid substitution at position 94. It has been previously demonstrated that the V94M protein is impaired relative to the wild-type enzyme predominantly at the level of V(max) rather than K(m). To address the molecular consequences the mutation imparts on the three-dimensional architecture of the enzyme, we have solved the structures of the V94M-substituted human epimerase complexed with NADH and UDP-glucose, UDP-galactose, UDP-GlcNAc, or UDP-GalNAc. In the wild-type enzyme, the hydrophobic side chain of Val(94) packs near the aromatic group of the catalytic Tyr(157) and serves as a molecular "fence" to limit the rotation of the glycosyl portions of the UDP-sugar substrates within the active site. The net effect of the V94M substitution is an opening up of the Ala(93) to Glu(96) surface loop, which allows free rotation of the sugars into nonproductive binding modes.
Organizational Affiliation:
Department of Biochemistry, University of Wisconsin, Madison, Wisconsin, 53706, USA.