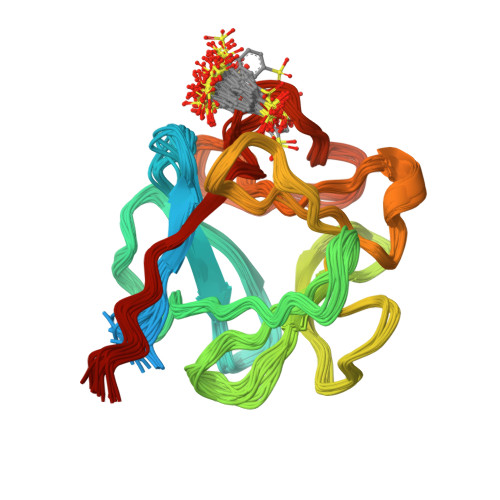
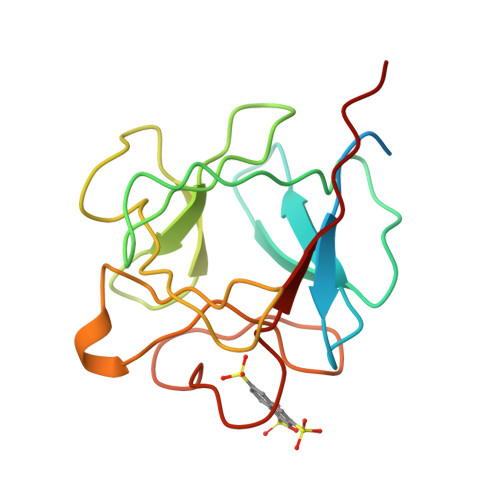
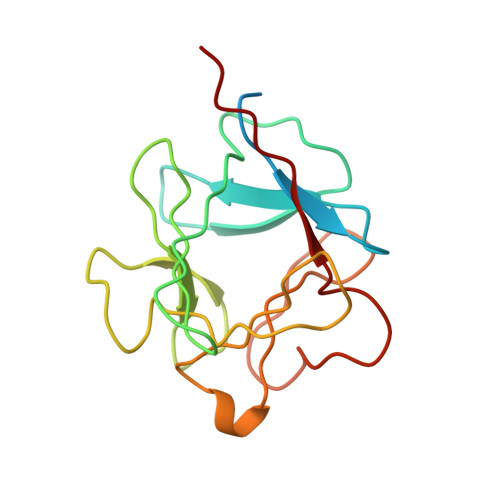
Solution structure of acidic fibroblast growth factor bound to 1,3, 6-naphthalenetrisulfonate: a minimal model for the anti-tumoral action of suramins and suradistas.
Lozano, R.M., Jimenez, M., Santoro, J., Rico, M., Gimenez-Gallego, G.(1998) J Mol Biology 281: 899-915
- PubMed: 9719643
- DOI: https://doi.org/10.1006/jmbi.1998.1977
- Primary Citation of Related Structures:
1RML - PubMed Abstract:
Recent data show that anti-angiogenesis may provide a promising route to treat cancer. Fibroblast growth factors (FGFs) are powerful angiogenic polypeptides, whose mitogenic activity requires the presence of heparin-like compounds. It has been shown that angiogenesis promoted by FGFs on inhibition by monoclonal antibodies and antisense targeting can also inhibit tumour growth. Derivatives of suramin, a polysulfonated binaphthyl urea and binaphthylsulfonated derivatives of distamycin, suradistas, constitute an important group of potential anti-cancer agents. These compounds compete with heparin in forming tight complexes with FGFs. This inhibits the recognition of these growth factors by their tyrosine kinase membrane receptors thereby suppressing their angiogenic activity. Here we show that 1,3,6-naphthalenetrisulfonate, a common chemical function of the suramins and suradistas with the highest anti-angiogenic activity inhibits the mitogenic activity of acidic fibroblast growth factor, and that this inhibition is relieved by increasing concentrations of heparin in the assay. We have also solved the three-dimensional structure in solution of the protein complexed to this compound. The structural data provide clues that may help in understanding the inhibitory effect of suramins and suradistas, and could contribute to the development of new anti-tumoral drugs.
Organizational Affiliation:
Centro de Investigaciones Biológicas (C.S.I.C.), Velázquez 144, Madrid, 28006, Spain.