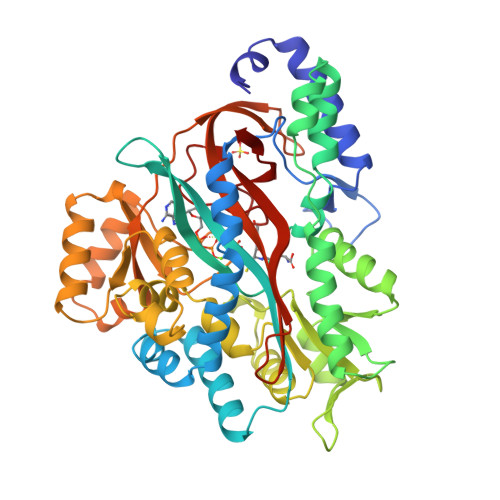
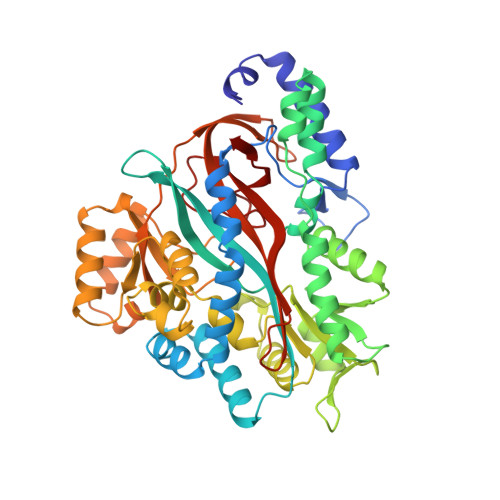
Molecular basis of glutathione synthetase deficiency and a rare gene permutation event.
Polekhina, G., Board, P.G., Gali, R.R., Rossjohn, J., Parker, M.W.(1999) EMBO J 18: 3204-3213
- PubMed: 10369661
- DOI: https://doi.org/10.1093/emboj/18.12.3204
- Primary Citation of Related Structures:
2HGS - PubMed Abstract:
Glutathione synthetase (GS) catalyses the production of glutathione from gamma-glutamylcysteine and glycine in an ATP-dependent manner. Malfunctioning of GS results in disorders including metabolic acidosis, 5-oxoprolinuria, neurological dysfunction, haemolytic anaemia and in some cases is probably lethal. Here we report the crystal structure of human GS (hGS) at 2.1 A resolution in complex with ADP, two magnesium ions, a sulfate ion and glutathione. The structure indicates that hGS belongs to the recently identified ATP-grasp superfamily, although it displays no detectable sequence identity with other family members including its bacterial counterpart, Escherichia coli GS. The difficulty in identifying hGS as a member of the family is due in part to a rare gene permutation which has resulted in a circular shift of the conserved secondary structure elements in hGS with respect to the other known ATP-grasp proteins. Nevertheless, it appears likely that the enzyme shares the same general catalytic mechanism as other ligases. The possibility of cyclic permutations provides an insight into the evolution of this family and will probably lead to the identification of new members. Mutations that lead to GS deficiency have been mapped onto the structure, providing a molecular basis for understanding their effects.
Organizational Affiliation:
The Ian Potter Foundation Protein Crystallography Laboratory, St Vincent's Institute of Medical Research, 41 Victoria Parade, Fitzroy, Victoria 3065, Australia.