Quaternary Structure of the Human Cdt1-Geminin Complex Regulates DNA Replication Licensing.
De Marco, V., Gillespie, P.J., Li, A., Karantzelis, N., Christodoulou, E., Klompmaker, R., Van Gerwen, S., Fish, A., Petoukhov, M.V., Iliou, M.S., Lygerou, Z., Medema, R.H., Blow, J.J., Svergun, D.I., Taraviras, S., Perrakis, A.(2009) Proc Natl Acad Sci U S A 106: 19807
- PubMed: 19906994
- DOI: https://doi.org/10.1073/pnas.0905281106
- Primary Citation of Related Structures:
2WVR - PubMed Abstract:
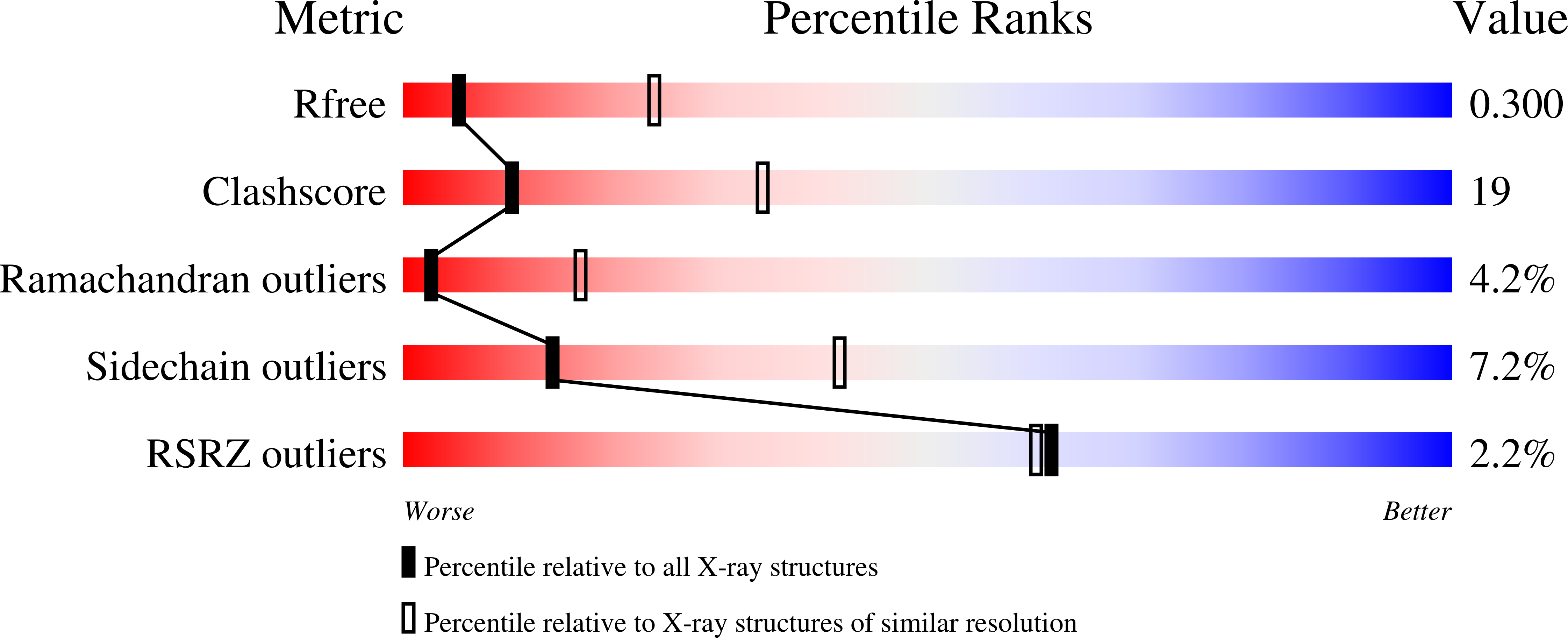
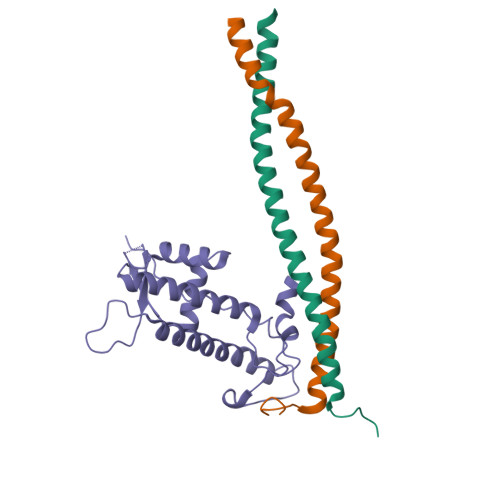
All organisms need to ensure that no DNA segments are rereplicated in a single cell cycle. Eukaryotes achieve this through a process called origin licensing, which involves tight spatiotemporal control of the assembly of prereplicative complexes (pre-RCs) onto chromatin. Cdt1 is a key component and crucial regulator of pre-RC assembly. In higher eukaryotes, timely inhibition of Cdt1 by Geminin is essential to prevent DNA rereplication. Here, we address the mechanism of DNA licensing inhibition by Geminin, by combining X-ray crystallography, small-angle X-ray scattering, and functional studies in Xenopus and mammalian cells. Our findings show that the Cdt1:Geminin complex can exist in two distinct forms, a "permissive" heterotrimer and an "inhibitory" heterohexamer. Specific Cdt1 residues, buried in the heterohexamer, are important for licensing. We postulate that the transition between the heterotrimer and the heterohexamer represents a molecular switch between licensing-competent and licensing-defective states.
Organizational Affiliation:
Department of Biochemistry, Netherlands Cancer Institute, Plesmanlaan 121, 1066CX Amsterdam, The Netherlands.