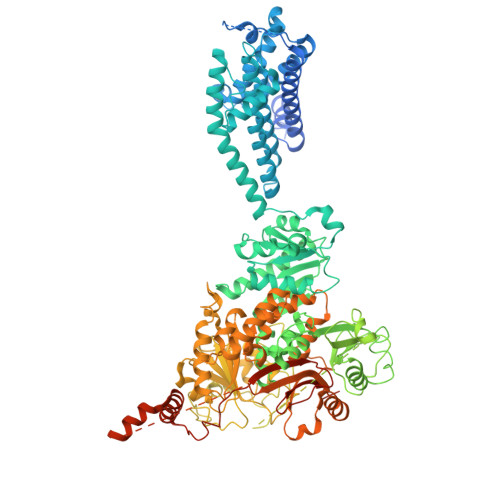
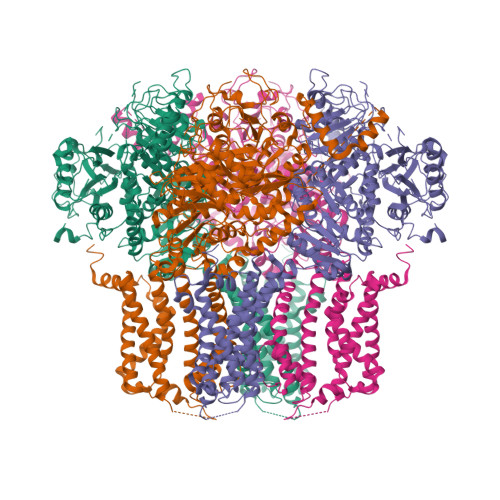
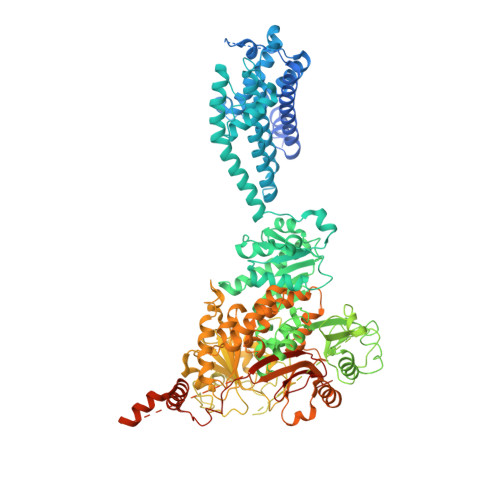
Structural Titration of Slo2.2, a Na(+)-Dependent K(+) Channel.
Hite, R.K., MacKinnon, R.(2017) Cell 168: 390-399.e11
- PubMed: 28111072
- DOI: https://doi.org/10.1016/j.cell.2016.12.030
- Primary Citation of Related Structures:
5U70, 5U76 - PubMed Abstract:
The stable structural conformations that occur along the complete reaction coordinate for ion channel opening have never been observed. In this study, we describe the equilibrium ensemble of structures of Slo2.2, a neuronal Na + -activated K + channel, as a function of the Na + concentration. We find that Slo2.2 exists in multiple closed conformations whose relative occupancies are independent of Na + concentration. An open conformation emerges from an ensemble of closed conformations in a highly Na + -dependent manner, without evidence of Na + -dependent intermediates. In other words, channel opening is a highly concerted, switch-like process. The midpoint of the structural titration matches that of the functional titration. A maximum open conformation probability approaching 1.0 and maximum functional open probability approaching 0.7 imply that, within the class of open channels, there is a subclass that is not permeable to ions.
Organizational Affiliation:
Rockefeller University and Howard Hughes Medical Institute, 1230 York Avenue, New York, NY 10065, USA.