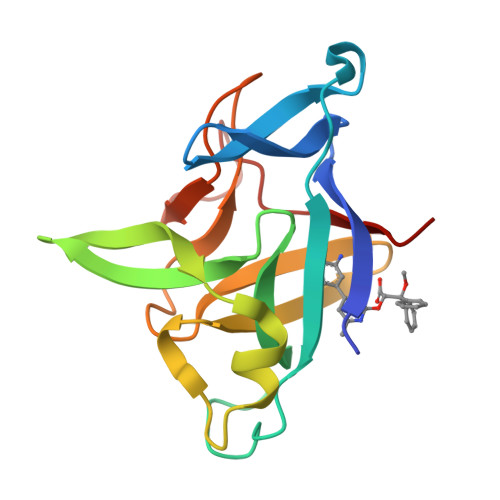
Small Molecule IL-36 gamma Antagonist as a Novel Therapeutic Approach for Plaque Psoriasis.
Todorovic, V., Su, Z., Putman, C.B., Kakavas, S.J., Salte, K.M., McDonald, H.A., Wetter, J.B., Paulsboe, S.E., Sun, Q., Gerstein, C.E., Medina, L., Sielaff, B., Sadhukhan, R., Stockmann, H., Richardson, P.L., Qiu, W., Argiriadi, M.A., Henry, R.F., Herold, J.M., Shotwell, J.B., McGaraughty, S.P., Honore, P., Gopalakrishnan, S.M., Sun, C.C., Scott, V.E.(2019) Sci Rep 9: 9089-9089
- PubMed: 31235749
- DOI: https://doi.org/10.1038/s41598-019-45626-w
- Primary Citation of Related Structures:
6P9E - PubMed Abstract:
IL-36 cytokines are pro-inflammatory members of the IL-1 family that are upregulated in inflammatory disorders. Specifically, IL-36γ is highly expressed in active psoriatic lesions and can drive pro-inflammatory processes in 3D human skin equivalents supporting a role for this target in skin inflammation. Small molecule antagonists of interleukins have been historically challenging to generate. Nevertheless, we performed a small molecule high-throughput screen to identify IL-36 antagonists using a novel TR-FRET binding assay. Several compounds, including 2-oxypyrimidine containing structural analogs of the marketed endothelin receptor A antagonist Ambrisentan, were identified as hits from the screen. A-552 was identified as a the most potent antagonist of human IL-36γ, but not the closely related family member IL-36α, was capable of attenuating IL-36γ induced responses in mouse and human disease models. Additionally, x-ray crystallography studies identified key amino acid residues in the binding pocket present in human IL-36γ that are absent in human IL-36α. A-552 represents a first-in-class small molecule antagonist of IL-36 signaling that could be used as a chemical tool to further investigate the role of this pathway in inflammatory skin diseases such as psoriasis.
Organizational Affiliation:
AbbVie Inc., 1 North Waukegan Rd., North Chicago, IL, 60064, USA. Viktor.Todorovic@abbvie.com.