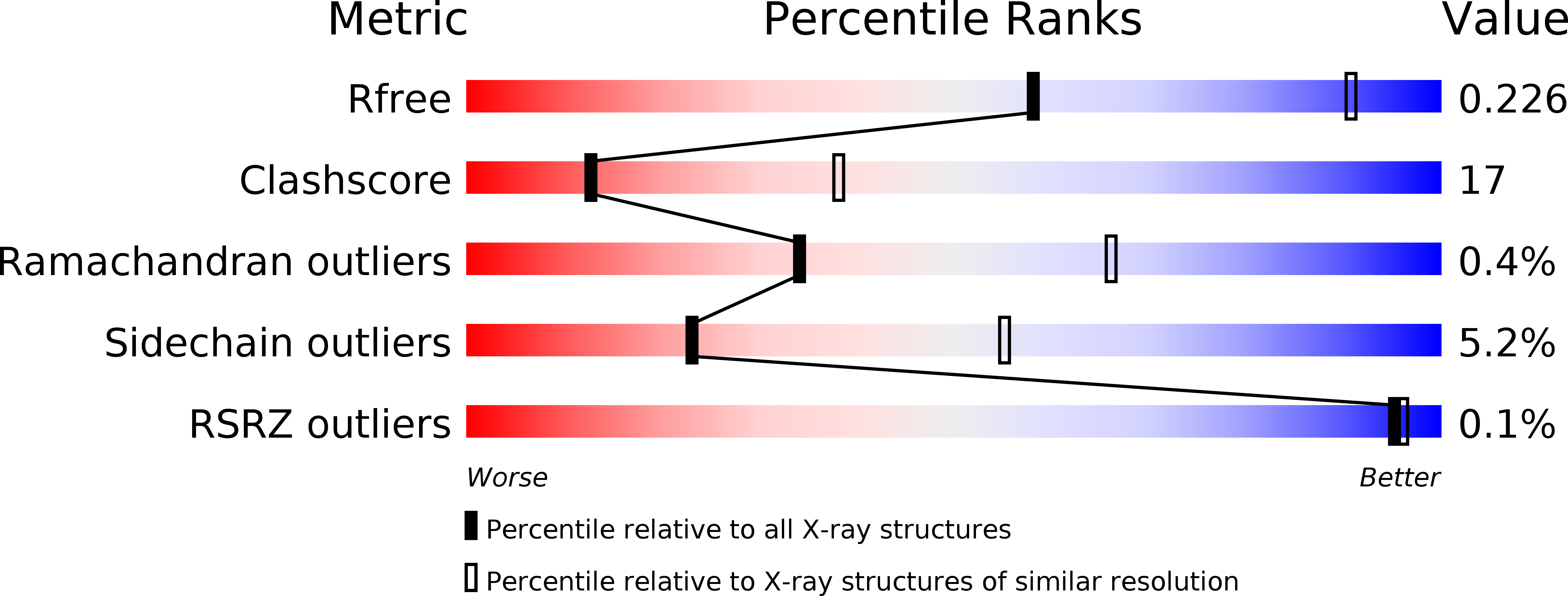
ADP-binding site of Escherichia coli succinyl-CoA synthetase revealed by x-ray crystallography.
Joyce, M.A., Fraser, M.E., James, M.N., Bridger, W.A., Wolodko, W.T.(2000) Biochemistry 39: 17-25
- PubMed: 10625475
- DOI: https://doi.org/10.1021/bi991696f
- Primary Citation of Related Structures:
1CQI, 1CQJ - PubMed Abstract:
Succinyl-CoA synthetase (SCS) catalyzes the following reversible reaction via a phosphorylated histidine intermediate (His 246alpha): succinyl-CoA + P(i) + NDP <--> succinate + CoA + NTP (N denotes adenosine or guanosine). To determine the structure of the enzyme with nucleotide bound, crystals of phosphorylated Escherichia coli SCS were soaked in successive experiments adopting progressive strategies. In the first experiment, 1 mM ADP (>15 x K(d)) was added; Mg(2+) ions were omitted to preclude the formation of an insoluble precipitate with the phosphate and ammonium ions. X-ray crystallography revealed that the enzyme was dephosphorylated, but the nucleotide did not remain bound to the enzyme (R(working) = 17.2%, R(free) = 22.8% for data to 2.9 A resolution). Catalysis requires Mg(2+) ions; hence, the "true" nucleotide substrate is probably an ADP-Mg(2+) complex. In the successful experiment, the phosphate buffer was exchanged with MOPS, the concentration of sulfate ions was lowered, and the concentrations of ADP and Mg(2+) ions were increased to 10.5 and 50 mM, respectively. X-ray diffraction data revealed an ADP-Mg(2+) complex bound in the ATP-grasp fold of the N-terminal domain of each beta-subunit (R(working) = 19.1%, R(free) = 24.7% for data to 3.3 A resolution). We describe the specific interactions of the nucleotide-Mg(2+) complex with SCS, compare these results with those for other proteins containing the ATP-grasp fold, and present a hypothetical model of the histidine-containing loop in the "down" position where it can interact with the nucleotide approximately 35 A from where His 246alpha is seen in both phosphorylated and dephosphorylated SCS.
Organizational Affiliation:
Department of Biochemistry, University of Alberta, Edmonton, Canada.