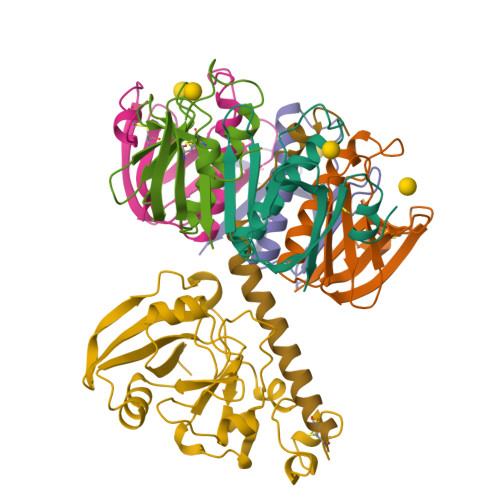
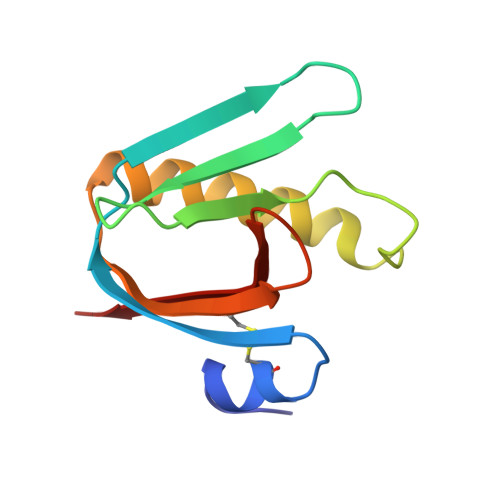
Galactose-binding site in Escherichia coli heat-labile enterotoxin (LT) and cholera toxin (CT).
Merritt, E.A., Sixma, T.K., Kalk, K.H., van Zanten, B.A., Hol, W.G.(1994) Mol Microbiol 13: 745-753
- PubMed: 7997185
- DOI: https://doi.org/10.1111/j.1365-2958.1994.tb00467.x
- Primary Citation of Related Structures:
1LTA - PubMed Abstract:
The galactose-binding site in cholera toxin and the closely related heat-labile enterotoxin (LT) from Escherichia coli is an attractive target for the rational design of potential anti-cholera drugs. In this paper we analyse the molecular structure of this binding site as seen in several crystal structures, including that of an LT:galactose complex which we report here at 2.2 A resolution. The binding surface on the free toxin contains several tightly associated water molecules and a relatively flexible loop consisting of residues 51-60 of the B subunit. During receptor binding this loop becomes tightly ordered by forming hydrogen bonds jointly to the GM1 pentasaccharide and to a set of water molecules which stabilize the toxin:receptor complex.
Organizational Affiliation:
Department of Biological Structure SM-20, University of Washington, Seattle 98195.