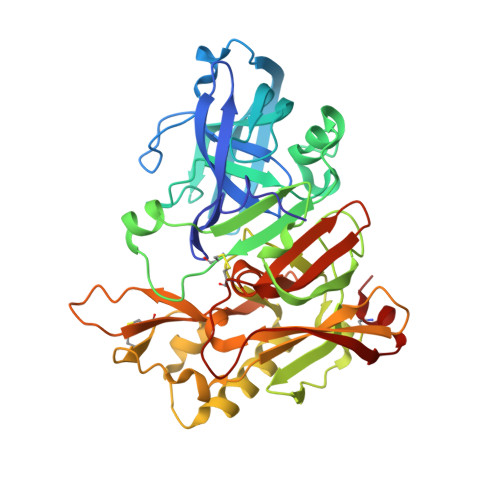
Structural locations and functional roles of new subsites S5, S6, and S7 in memapsin 2 (beta-secretase).
Turner III, R.T., Hong, L., Koelsch, G., Ghosh, A.K., Tang, J.(2005) Biochemistry 44: 105-112
- PubMed: 15628850
- DOI: https://doi.org/10.1021/bi048106k
- Primary Citation Related Structures:
1XN2, 1XN3 - PubMed Abstract:
Memapsin 2 (beta-secretase) is the membrane-anchored aspartic protease that initiates the cleavage of beta-amyloid precursor protein (APP), leading to the production of amyloid-beta (Abeta), a major factor in the pathogenesis of Alzheimer's disease. The active site of memapsin 2 has been shown, with kinetic data and crystal structures, to bind to eight substrate residues (P(4)-P(4)'). We describe here that the addition of three substrate residues from P(7) to P(5) strongly influences the hydrolytic activity by memapsin 2 and these subsites prefer hydrophobic residues, especially tryptophan. A crystal structure of memapsin 2 complexed with a statine-based inhibitor spanning P(10)-P(4)' revealed the binding positions of P(5)-P(7) residues. Kinetic studies revealed that the addition of these substrate residues contributes to the decrease in K(m) and increase in k(cat) values, suggesting that these residues contribute to both substrate recognition and transition-state binding. The crystal structure of a new inhibitor, OM03-4 (K(i) = 0.03 nM), bound to memapsin 2 revealed the interaction of a tryptophan with the S(6) subsite of the protease.
- Protein Studies Program, Oklahoma Medical Research Foundation, Department of Biochemistry and Molecular Biology, University of Oklahoma Health Science Center, Oklahoma City, Oklahoma 73104, USA.
Organizational Affiliation: