Crystal structure of TTK kinase domain with an inhibitor: 401348
Qiu, W., Plotnikov, A.N., Plotnikova, O., Feher, M., Awrey, D.E., Chirgadze, N.Y.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Dual specificity protein kinase TTK | 281 | Homo sapiens | Mutation(s): 0 Gene Names: TTK, MPS1, MPS1L1 EC: 2.7.12.1 | 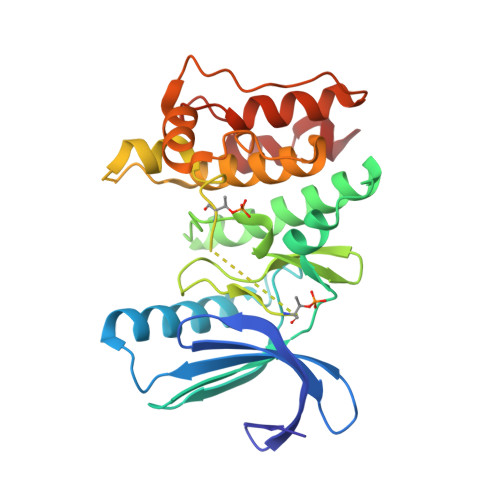 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P33981 GTEx: ENSG00000112742 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P33981 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 1PF Download:Ideal Coordinates CCD File | B [auth A] | 4-(cyclohexylmethoxy)-3-{4-[(1-methylpiperidin-4-yl)oxy]phenyl}-2H-indazole C26 H33 N3 O2 ZXTGHNQQRVHEIZ-UHFFFAOYSA-N |  | ||
| PEG Download:Ideal Coordinates CCD File | E [auth A], F [auth A] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | C [auth A], D [auth A] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Download:Ideal Coordinates CCD File | G [auth A], H [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| TPO Query on TPO | A | L-PEPTIDE LINKING | C4 H10 N O6 P |  | THR |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 70.704 | α = 90 |
| b = 107.939 | β = 90 |
| c = 113.233 | γ = 90 |
| Software Name | Purpose |
|---|---|
| JDirector | data collection |
| PHASER | phasing |
| BUSTER | refinement |
| XDS | data reduction |
| XDS | data scaling |