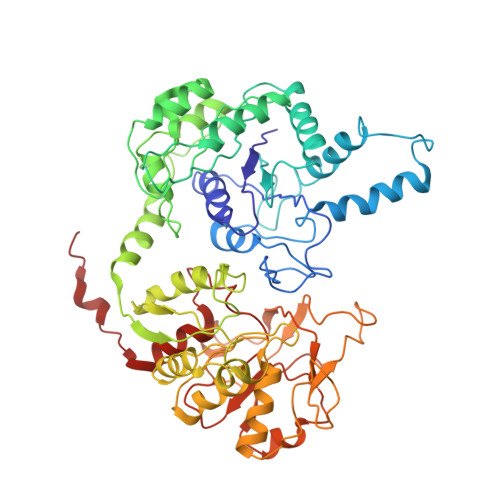
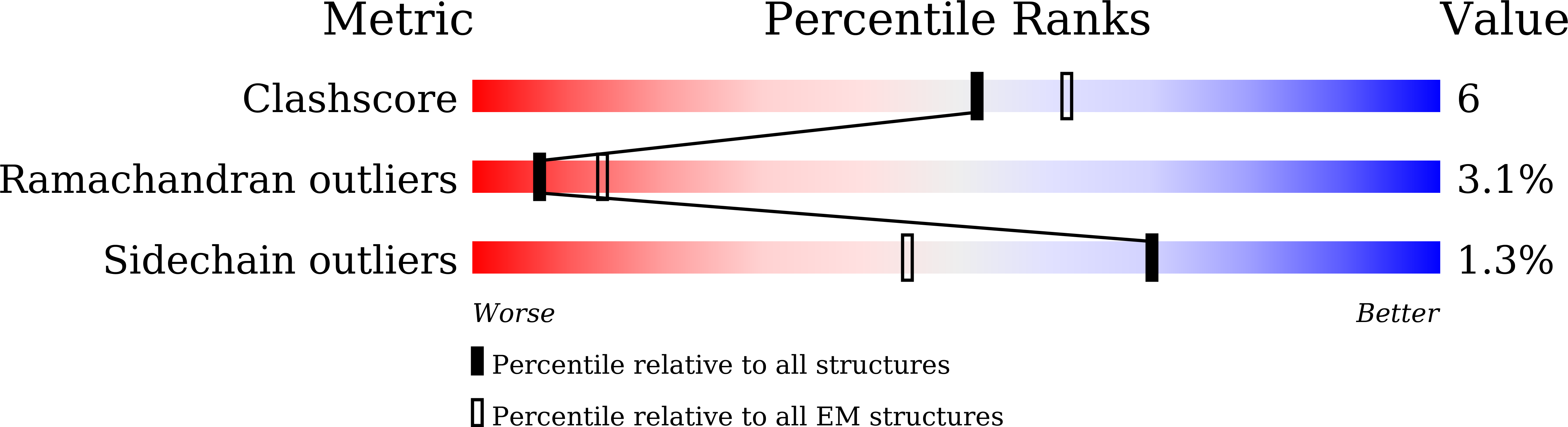Drosophila CTP synthase can form distinct substrate- and product-bound filaments.
Zhou, X., Guo, C.J., Hu, H.H., Zhong, J., Sun, Q., Liu, D., Zhou, S., Chang, C.C., Liu, J.L.(2019) J Genet Genomics 46: 537-545
- PubMed: 31902586
- DOI: https://doi.org/10.1016/j.jgg.2019.11.006
- Primary Citation of Related Structures:
6L6Z, 6LFG - PubMed Abstract:
Intracellular compartmentation is a key strategy for the functioning of a cell. In 2010, several studies revealed that the metabolic enzyme CTP synthase (CTPS) can form filamentous structures termed cytoophidia in prokaryotic and eukaryotic cells. However, recent structural studies showed that CTPS only forms inactive product-bound filaments in bacteria while forming active substrate-bound filaments in eukaryotic cells. In this study, using negative staining and cryo-electron microscopy, we demonstrate that Drosophila CTPS, whether in substrate-bound or product-bound form, can form filaments. Our results challenge the previous model and indicate that substrate-bound and product-bound filaments can coexist in the same species. We speculate that the ability to switch between active and inactive cytoophidia in the same cells provides an additional layer of metabolic regulation.
Organizational Affiliation:
School of Life Science and Technology, ShanghaiTech University, Shanghai, 201210, China.