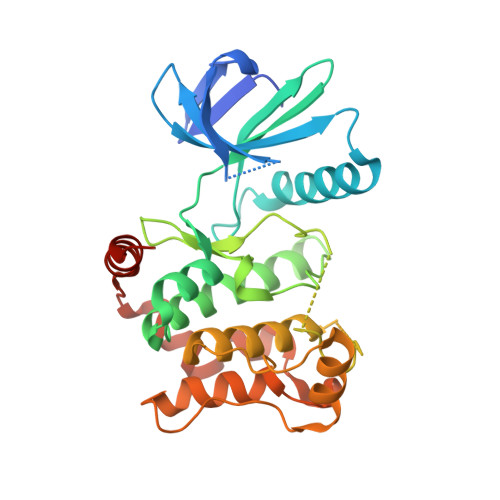
Characterization of Covalent Pyrazolopyrimidine-MKK7 Complexes and a Report on a Unique DFG-in/Leu-in Conformation of Mitogen-Activated Protein Kinase Kinase 7 (MKK7).
Wolle, P., Engel, J., Smith, S., Goebel, L., Hennes, E., Lategahn, J., Rauh, D.(2019) J Med Chem 62: 5541-5546
- PubMed: 31083997
- DOI: https://doi.org/10.1021/acs.jmedchem.9b00472
- Primary Citation Related Structures:
6QFL, 6QFR, 6QFT, 6QG4, 6QG7, 6QHO, 6QHR - PubMed Abstract:
Pyrazolopyrimidines are well-established as covalent inhibitors of protein kinases such as the epidermal growth factor receptor or Bruton's tyrosine kinase, and we recently described their potential in targeting mitogen-activated protein kinase kinase 7 (MKK7). Herein, we report the structure-activity relationship of pyrazolopyrimidine-based MKK7 inhibitors and solved several complex crystal structures to gain insights into their binding mode. In addition, we present two structures of apo-MKK7, exhibiting a DFG-out and an unprecedented DFG-in/Leu-in conformation.
- Faculty of Chemistry and Chemical Biology , TU Dortmund University , Otto-Hahn-Strasse 4a , 44227 Dortmund , Germany.
Organizational Affiliation: