Difluorophenyl diacylhydrazides: Potent inhibitors of Serum- and Glucocorticoid-inducible Kinase 1 (SGK1)
Graedler, U.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| cAMP-dependent protein kinase catalytic subunit alpha | 351 | Homo sapiens | Mutation(s): 0 Gene Names: PRKACA, PKACA EC: 2.7.11.11 | 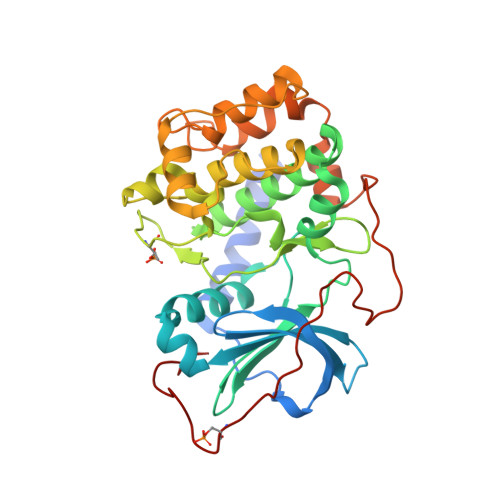 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P17612 GTEx: ENSG00000072062 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P17612 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| cAMP-dependent protein kinase inhibitor alpha | 20 | Homo sapiens | Mutation(s): 0 Gene Names: PKIA, PRKACN1 | 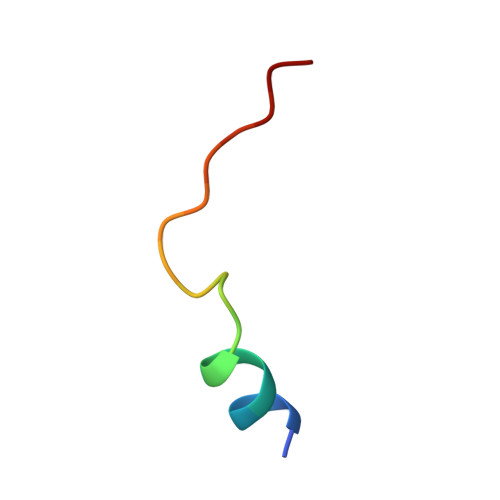 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P61925 GTEx: ENSG00000171033 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P61925 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| J4B Download:Ideal Coordinates CCD File | C [auth A] | ~{N}'-[(2~{S})-2-[3,5-bis(fluoranyl)phenyl]-2-oxidanyl-ethanoyl]-2-ethyl-3-methyl-4-oxidanyl-benzohydrazide C18 H18 F2 N2 O4 SSNAPUUWBPZGOY-INIZCTEOSA-N |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| SEP Query on SEP | A | L-PEPTIDE LINKING | C3 H8 N O6 P |  | SER |
| TPO Query on TPO | A | L-PEPTIDE LINKING | C4 H10 N O6 P |  | THR |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 72.359 | α = 90 |
| b = 75.881 | β = 90 |
| c = 79.968 | γ = 90 |
| Software Name | Purpose |
|---|---|
| BUSTER | refinement |
| TRUNCATE | data reduction |
| autoPROC | data scaling |
| MOLREP | phasing |