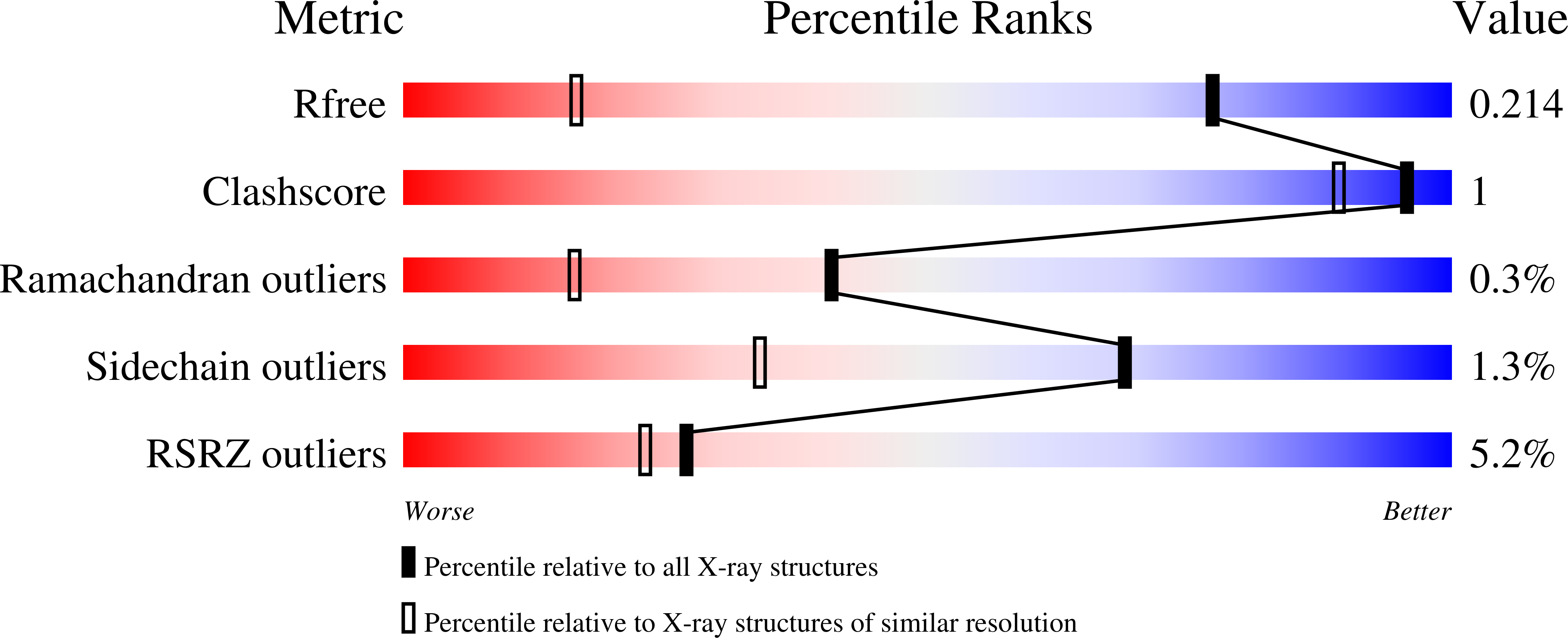
A fragment-based approach leading to the discovery of inhibitors of CK2 alpha with a novel mechanism of action.
Brear, P., De Fusco, C., Atkinson, E.L., Iegre, J., Francis-Newton, N.J., Venkitaraman, A.R., Hyvonen, M., Spring, D.R.(2022) RSC Med Chem 13: 1420-1426
- PubMed: 36426237
- DOI: https://doi.org/10.1039/d2md00161f
- Primary Citation of Related Structures:
7ZY0, 7ZY2, 7ZY5, 7ZY8, 7ZYD, 7ZYK, 7ZYO, 7ZYR, 8AE7, 8AEC, 8AEK, 8AEM - PubMed Abstract:
CK2 is a ubiquitous protein kinase with an anti-apoptotic role and is found to be overexpressed in multiple cancer types. To this end, the inhibition of CK2 is of great interest with regard to the development of novel anti-cancer therapeutics. ATP-site inhibition of CK2 is possible; however, this typically results in poor selectivity due to the highly conserved nature of the catalytic site amongst kinases. An alternative methodology for the modulation of CK2 activity is through allosteric inhibition. The recently identified αD site represents a promising binding site for allosteric inhibition of CK2α. The work presented herein describes the development of a series of CK2α allosteric inhibitors through iterative cycles of X-ray crystallography and enzymatic assays, in addition to both fragment growing and fragment merging design strategies. The lead fragment developed, fragment 8, exhibits a high ligand efficiency, displays no drop off in activity between enzymatic and cellular assays, and successfully engages CK2α in cells. Furthermore, X-ray crystallographic analysis provided indications towards a novel mechanism of allosteric inhibition through αD site binding. Fragments described in this paper therefore represent promising starting points for the development of highly selective allosteric CK2 inhibitors.
Organizational Affiliation:
Department of Biochemistry, University of Cambridge Tennis Court Road CB2 1GA Cambridge UK mh256@cam.ac.uk.