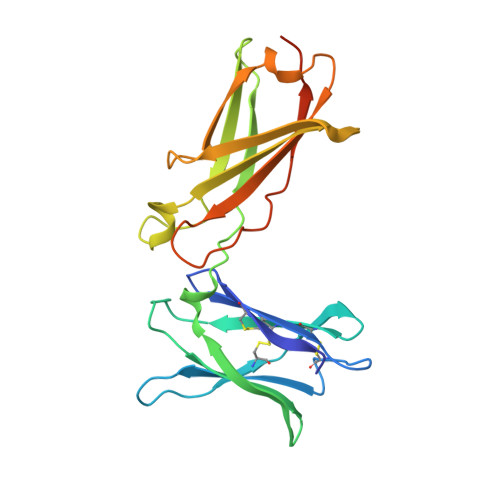
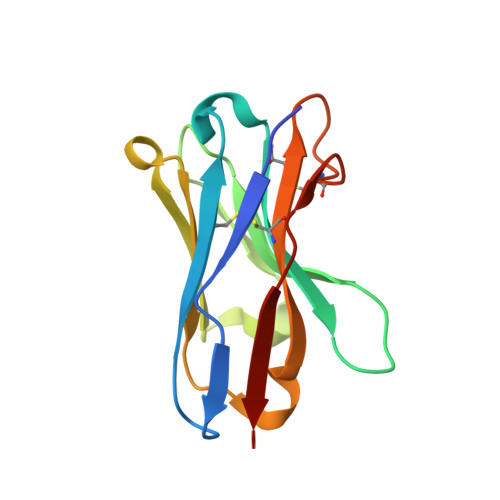
A novel inhalable nanobody targeting IL-4R alpha for the treatment of asthma.
Zhu, M., Ma, L., Zhong, P., Huang, J., Gai, J., Li, G., Li, Y., Qiao, P., Gu, H., Li, X., Yin, Y., Zhang, L., Deng, Z., Sun, B., Chen, Z., Ding, Y., Wan, Y.(2024) J Allergy Clin Immunol 154: 1008-1021
- PubMed: 38871183
- DOI: https://doi.org/10.1016/j.jaci.2024.05.027
- Primary Citation of Related Structures:
8K4Q - PubMed Abstract:
Inhalable biologics represent a promising approach to improve the efficacy and safety of asthma treatment. Although several monoclonal antibodies (mAbs) targeting IL-4Rα have been approved or are undergoing clinical trials, the development of inhalable mAbs targeting IL-4Rα presents significant challenges. Capitalizing on the distinctive advantages of nanobodies (Nbs) in maintaining efficacy during storage and administration, we sought to develop a novel inhalable IL-4Rα Nb for effectively treating asthma. Three IL-4Rα immunized Nb libraries were utilized to generate specific and functional IL-4Rα Nbs. LQ036, a bivalent Nb comprising two HuNb103 units, was constructed with a high affinity and specificity for hIL-4Rα. The efficacy, pharmacokinetic and safety of inhaled LQ036 were evaluated in B-hIL4/hIL4Ra humanized mice. LQ036 inhibited secreted embryonic alkaline phosphatase (SEAP) reporter activity, TF-1 cell proliferation, and suppressed pSTAT6 in T cells from asthma patients. Crystal structure analysis revealed a binding region similar to Dupilumab but with higher affinity, leading to better efficacy in blocking the signaling pathway. HuNb103 competed with IL-4 and IL-13 for IL-4Rα binding. Additionally, LQ036 significantly inhibited OVA-specific IgE levels in serum, CCL17 levels in BALF, bronchial mucous cell hyperplasia, and airway goblet cell hyperplasia in B-hIL4/hIL4Ra humanized mice. Inhaled LQ036 exhibited favorable pharmacokinetics, safety and tissue distribution, with higher concentrations observed in the lungs and bronchi. These findings from preclinical studies establish the safety and efficacy of inhaled LQ036, underscoring its potential as a pioneering inhalable biologic therapy for asthma.
- Shanghai Novamab Biopharmaceuticals Co., Ltd., Shanghai 201318, China.
Organizational Affiliation: