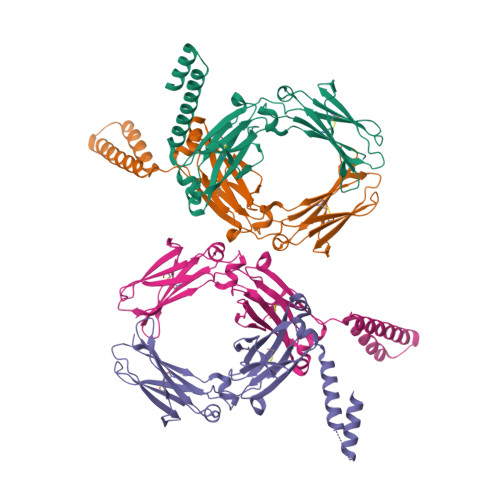
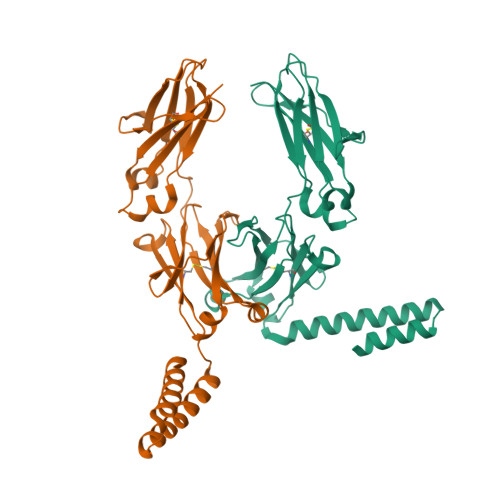
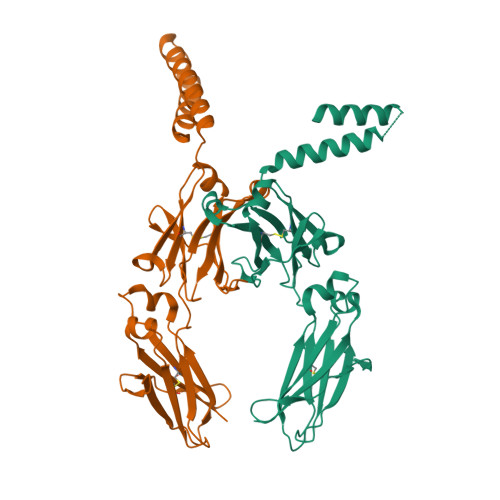
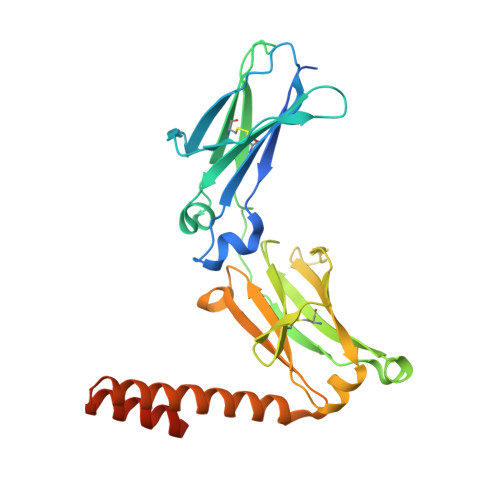
A human histidyl-tRNA synthetase splice variant therapeutic targets NRP2 to resolve lung inflammation and fibrosis.
Nangle, L.A., Xu, Z., Siefker, D., Burkart, C., Chong, Y.E., Zhai, L., Geng, Y., Polizzi, C., Guy, L., Eide, L., Tong, Y., Klopp-Savino, S., Ferrer, M., Rauch, K., Wang, A., Hamel, K., Crampton, S., Paz, S., Chiang, K.P., Do, M.H., Burman, L., Lee, D., Zhang, M., Ogilvie, K., King, D., Adams, R.A., Schimmel, P.(2025) Sci Transl Med 17: eadp4754-eadp4754
- PubMed: 40073151
- DOI: https://doi.org/10.1126/scitranslmed.adp4754
- Primary Citation of Related Structures:
8YOR, 8YP1 - PubMed Abstract:
Interstitial lung disease (ILD) consists of a group of immune-mediated disorders that can cause inflammation and progressive fibrosis of the lungs, representing an area of unmet medical need given the lack of disease-modifying therapies and toxicities associated with current treatment options. Tissue-specific splice variants (SVs) of human aminoacyl-tRNA synthetases (aaRSs) are catalytic nulls thought to confer regulatory functions. One example from human histidyl-tRNA synthetase (HARS), termed HARS WHEP because the splicing event resulted in a protein encompassing the WHEP-TRS domain of HARS (a structurally conserved domain found in multiple aaRSs), is enriched in human lung and up-regulated by inflammatory cytokines in lung and immune cells. Structural analysis of HARS WHEP confirmed a well-organized helix-turn-helix motif. This motif bound specifically and selectively to neuropilin-2 (NRP2), a receptor expressed by myeloid cells in active sites of inflammation, to inhibit expression of proinflammatory receptors and cytokines and to down-regulate inflammatory pathways in primary human macrophages. In animal models of lung injury and ILD, including bleomycin treatment, silicosis, sarcoidosis, chronic hypersensitivity pneumonitis, systemic sclerosis, and rheumatoid arthritis-ILD, HARS WHEP reduced lung inflammation, immune cell infiltration, and fibrosis. In patients with sarcoidosis, efzofitimod treatment resulted in down-regulation of gene expression for inflammatory pathways in peripheral immune cells and stabilization of inflammatory biomarkers in serum after steroid tapering. We demonstrate the immunomodulatory activity of HARS WHEP and present preclinical data supporting ongoing clinical development of the biologic efzofitimod based on HARS WHEP in ILD.
Organizational Affiliation:
aTyr Pharma, San Diego, CA 92121, USA.