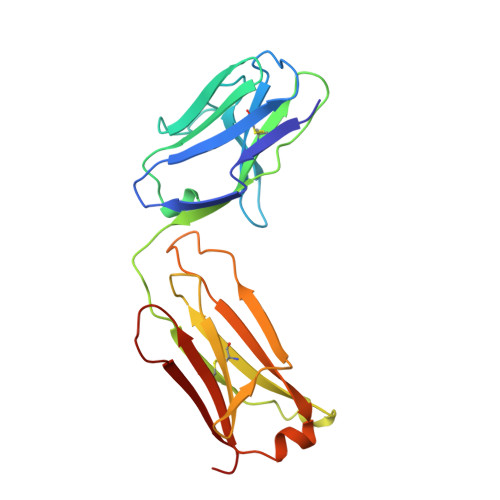
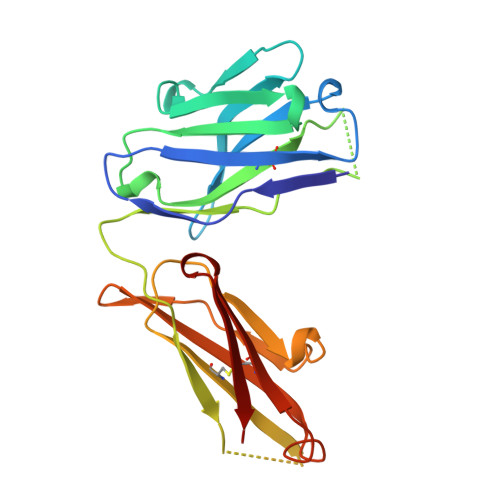
A comparison of the crystallographic structures of two catalytic antibodies with esterase activity.
Buchbinder, J.L., Stephenson, R.C., Scanlan, T.S., Fletterick, R.J.(1998) J Mol Biology 282: 1033-1041
- PubMed: 9753552 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1998.2025
- Primary Citation Related Structures:
1A0Q - PubMed Abstract:
The crystallographic structure of the Fab fragment of the catalytic antibody, 29G11, complexed with an (S)-norleucine phenyl phosphonate transition state analog was determined at 2.2 A resolution. The antibody catalyzes the hydrolysis of norleucine phenyl ester with (S)-enantioselectivity. The shape and charge complementarity of the binding pocket for the hapten account for the preferential binding of the (S)-enantiomer of the substrate. The structure is compared to that of the more catalytically efficient antibody, 17E8, induced by the same hapten transition state analog. 29G11 has different residues from 17E8 at eight positions in the heavy chain, including four substitutions in the hapten-binding pocket: A33V, S95G, S99R and Y100AN, and four substitutions at positions remote from the catalytic site, I28T, R40K, V65G and F91L. The two antibodies show large differences in the orientations of their variable and constant domains, reflected by a 32 degrees difference in their elbow angles. The VL and VH domains in the two antibodies differ by a rotation of 8.8 degrees. The hapten binds in similar orientations and locations in 29G11 and 17E8, which appear to have catalytic groups in common, though the changes in the association of the variable domains affect the precise positioning of residues in the hapten-binding pocket.
- Department of Biochemistry and Biophysics, University of California, San Francisco, San Francisco, CA, 94143-0448, USA.
Organizational Affiliation: