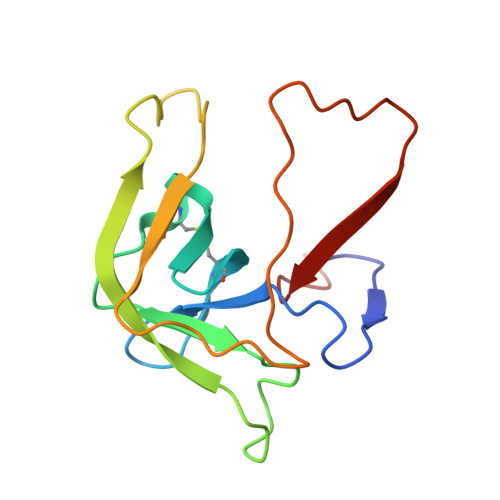
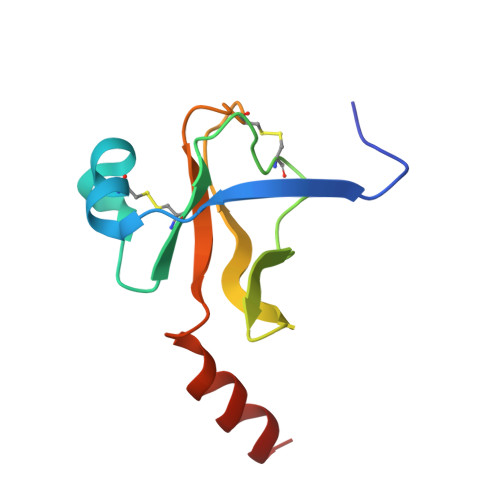
Refined crystal structures of "aged" and "non-aged" organophosphoryl conjugates of gamma-chymotrypsin.
Harel, M., Su, C.T., Frolow, F., Ashani, Y., Silman, I., Sussman, J.L.(1991) J Mol Biology 221: 909-918
- PubMed: 1942036 Search on PubMed
- DOI: https://doi.org/10.1016/0022-2836(91)80183-u
- Primary Citation Related Structures:
1GCD, 1GMH - PubMed Abstract:
"Aged" organophosphoryl conjugates of serine hydrolases differ from the corresponding "non-aged" conjugates in their striking resistance to nucleophilic reactivation. The refined X-ray structures of "aged" and "non-aged" organophosphoryl conjugates of gamma-chymotrypsin were compared in order to understand the molecular basis for this resistance of "aged" conjugates. "Aged" and "non-aged" crystalline organophosphoryl-gamma-chymotrypsin conjugates were obtained by prolonged soaking of native gamma-chymotrypsin crystals with appropriate organophosphates. Thus, a representative "non-aged" conjugate, diethylphosphoryl-gamma-chymotrypsin, was obtained by soaking native crystals with paraoxon (diethyl-p-nitrophenyl phosphate), and a closely related "aged" conjugate, monoisopropyl-gamma-chymotrypsin, was obtained by soaking with diisopropylphosphorofluoridate. In both crystalline conjugates, the refined structures clearly reveal a high occupancy of the active site by the appropriate organophosphoryl moiety within covalent bonding distance of Ser195 O gamma. Whereas in the "non-aged" conjugate both ethyl groups can be visualized clearly, in the putative "aged" conjugate, as expected, only one isopropyl group is present. There is virtually no difference between the "aged" and "non-aged" conjugates either with respect to the conformation of the polypeptide backbone as a whole or with respect to the positioning of the side-chains within the active site. In the "aged" conjugate, however, close proximity (2.6 A) of the negatively charged phosphate oxygen atom of the dealkylated organophosphoryl group to His57 N epsilon 2 indicates the presence of a salt bridge between these two moieties. In contrast, in the "non-aged" conjugate the DEP moiety retains its two alkyl groups; thus, lacking a negative oxygen atom, it does not enter into such a charge-charge interaction and its nearest oxygen atom is 3.6 A away from His57 N epsilon 2. It is suggested that steric constraints imposed by the salt bridge in the "aged" conjugate lie at the basis of its resistance to reactivation.
- Department of Structural Chemistry, Weizmann Institute of Science, Rehovot, Israel.
Organizational Affiliation: