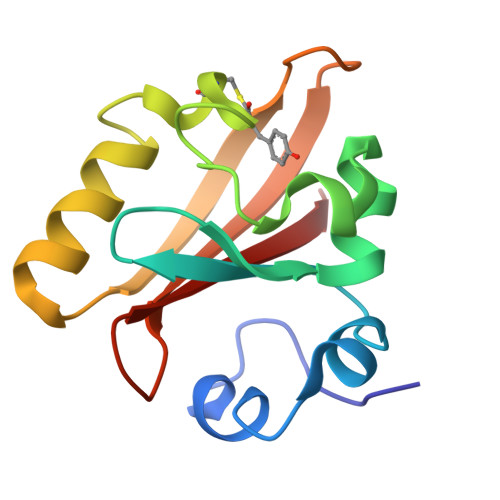
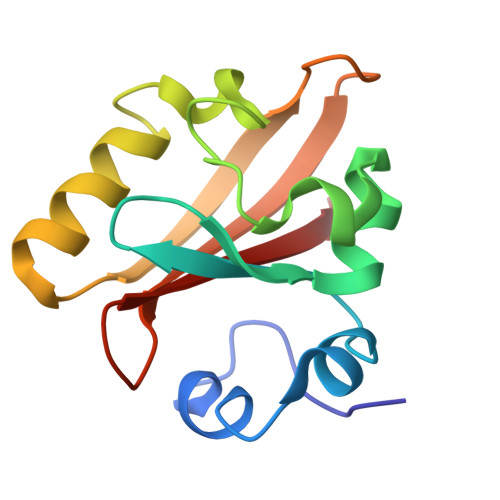
A Structural Pathway for Signaling in the E46Q Mutant of Photoactive Yellow Protein
Rajagopal, S., Anderson, S., Srajer, V., Schmidt, M., Pahl, R., Moffat, K.(2005) Structure 13: 55-63
- PubMed: 15642261
- DOI: https://doi.org/10.1016/j.str.2004.10.016
- Primary Citation of Related Structures:
1T18, 1T19, 1T1A, 1T1B, 1T1C - PubMed Abstract:
In the bacterial photoreceptor photoactive yellow protein (PYP), absorption of blue light by its chromophore leads to a conformational change in the protein associated with differential signaling activity, as it executes a reversible photocycle. Time-resolved Laue crystallography allows structural snapshots (as short as 150 ps) of high crystallographic resolution (approximately 1.6 A) to be taken of a protein as it functions. Here, we analyze by singular value decomposition a comprehensive time-resolved crystallographic data set of the E46Q mutant of PYP throughout the photocycle spanning 10 ns-100 ms. We identify and refine the structures of five distinct intermediates and provide a plausible chemical kinetic mechanism for their inter conversion. A clear structural progression is visible in these intermediates, in which a signal generated at the chromophore propagates through a distinct structural pathway of conserved residues and results in structural changes near the N terminus, over 20 A distant from the chromophore.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology, University of Chicago, Illinois 60637, USA.