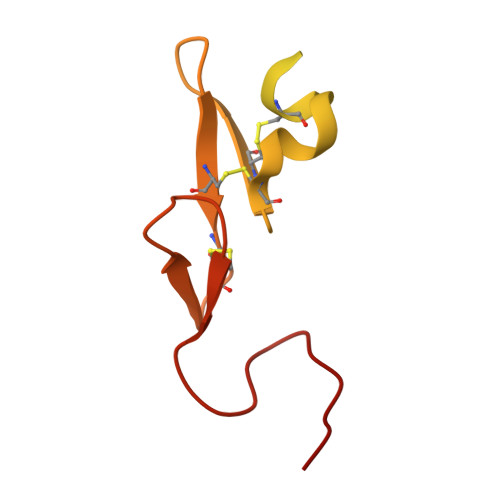Design and Synthesis of Orally Active Pyrrolidin-2-One-Based Factor Xa Inhibitors
Watson, N.S., Brown, D., Campbell, M., Chan, C., Chaudry, L., Convery, M.A., Fenwick, R., Hamblin, J.N., Haslam, C., Kelly, H.A., King, N.P., Kurtis, C.L., Leach, A.R., Manchee, G.R., Mason, A.M., Mitchell, C., Patel, C., Patel, V.K., Senger, S., Shah, G.P., Weston, H.E., Whitworth, C., Young, R.J.(2006) Bioorg Med Chem Lett 16: 3784
- PubMed: 16697194 Search on PubMed
- DOI: https://doi.org/10.1016/j.bmcl.2006.04.053
- Primary Citation Related Structures:
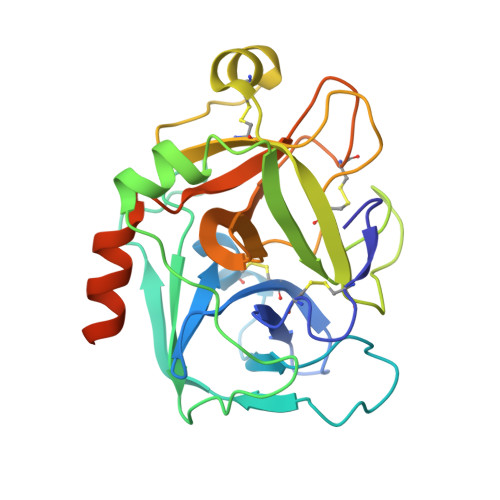
2CJI - PubMed Abstract:
A series of novel, non-basic 3-(6-chloronaphth-2-ylsulfonyl)aminopyrrolidin-2-one-based factor Xa (fXa) inhibitors, incorporating an alanylamide P4 group, was designed and synthesised. Within this series, the N-2-(morpholin-4-yl)-2-oxoethyl derivative 24 was shown to be a potent, selective fXa inhibitor with good anticoagulant activity. Moreover, 24 possessed highly encouraging rat and dog pharmacokinetic profiles with excellent oral bioavailabilities in both species.
- GlaxoSmithKline, Medicines Research Centre, Gunnels Wood Road, Stevenage, Hertfordshire SG1 2NY, UK. Nigel.S.Watson@gsk.com
Organizational Affiliation: