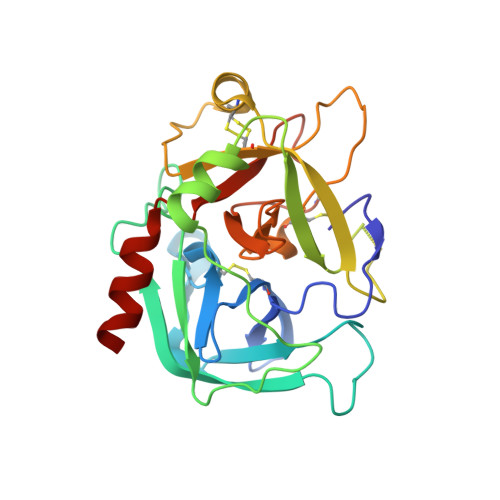
Crystal structure of thrombin in a self-inhibited conformation.
Pineda, A.O., Chen, Z.W., Bah, A., Garvey, L.C., Mathews, F.S., Di Cera, E.(2006) J Biological Chem 281: 32922-32928
- PubMed: 16954215 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M605530200
- Primary Citation Related Structures:
2GP9 - PubMed Abstract:
The activating effect of Na(+) on thrombin is allosteric and depends on the conformational transition from a low activity Na(+)-free (slow) form to a high activity Na(+)-bound (fast) form. The structures of these active forms have been solved. Recent structures of thrombin obtained in the absence of Na(+) have also documented inactive conformations that presumably exist in equilibrium with the active slow form. The validity of these inactive slow form structures, however, is called into question by the presence of packing interactions involving the Na(+) site and the active site regions. Here, we report a 1.87A resolution structure of thrombin in the absence of inhibitors and salts with a single molecule in the asymmetric unit and devoid of significant packing interactions in regions involved in the allosteric slow --> fast transition. The structure shows an unprecedented self-inhibited conformation where Trp-215 and Arg-221a relocate >10A to occlude the active site and the primary specificity pocket, and the guanidinium group of Arg-187 penetrates the protein core to fill the empty Na(+)-binding site. The extreme mobility of Trp-215 was investigated further with the W215P mutation. Remarkably, the mutation significantly compromises cleavage of the anticoagulant protein C but has no effect on the hydrolysis of fibrinogen and PAR1. These findings demonstrate that thrombin may assume an inactive conformation in the absence of Na(+) and that its procoagulant and anticoagulant activities are closely linked to the mobility of residue 215.
- Department of Biochemistry and Molecular Biophysics, Washington University School of Medicine, St. Louis, MO 63110, USA.
Organizational Affiliation: