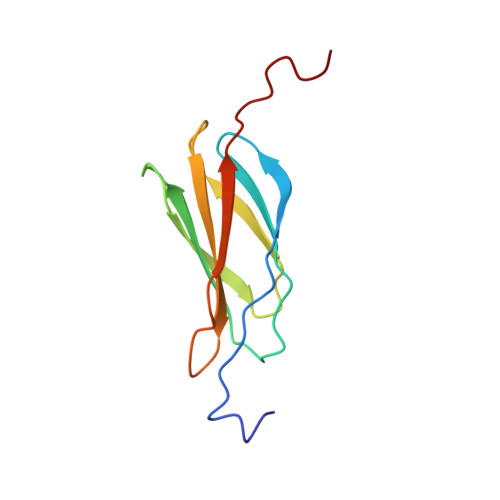
Structure and Unfolding of the Third Type III Domain from Human Fibronectin.
Stine, J.M., Sun, Y., Armstrong, G., Bowler, B.E., Briknarova, K.(2015) Biochemistry 54: 6724-6733
- PubMed: 26517579 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/acs.biochem.5b00818
- Primary Citation Related Structures:
2N1K - PubMed Abstract:
Fibronectin is a modular extracellular matrix protein that is essential for vertebrate development. The third type III domain (3FN3) in fibronectin interacts with other parts of fibronectin and with anastellin, a protein fragment that causes fibronectin aggregation. 3FN3 opens readily both as an isolated domain in solution and when part of fibronectin in stretched fibrils, and it was proposed that this opening is important for anastellin binding. We determined the structure of 3FN3 using nuclear magnetic resonance spectroscopy, and we investigated its stability, folding, and unfolding. Similar to most other FN3 domains, 3FN3 contains two antiparallel β-sheets that are composed of three (A, B, and E) and four (C, D, F, and G) β-strands, respectively, and are held together by a conserved hydrophobic interface. cis-trans isomerization of P847 at the end of β-strand C leads to observable conformational heterogeneity in 3FN3, with a cis peptide bond present in almost one-quarter of the molecules. The chemical stability of 3FN3 is relatively low, but the folding rate constant in the absence of denaturant is in the same range as those of other, more stable FN3 domains. Interestingly, the unfolding rate constant in the absence of denaturant is several orders of magnitude higher than the unfolding rate constants of other FN3 domains investigated to date. This unusually fast rate is comparable to the rate of binding of 3FN3 to anastellin at saturating anastellin concentrations, consistent with the model in which 3FN3 has to unfold to interact with anastellin.
- Department of Chemistry and Biochemistry, University of Montana , Missoula, Montana 59812, United States.
Organizational Affiliation: