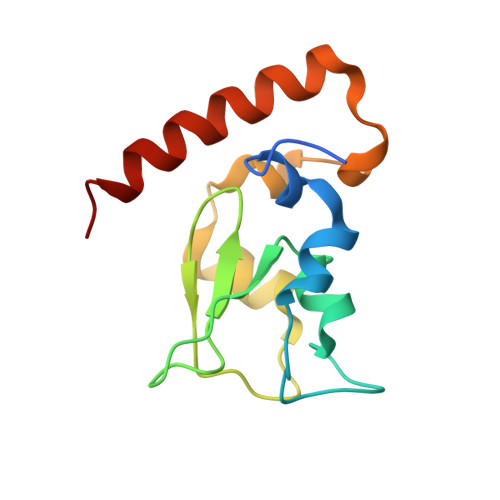
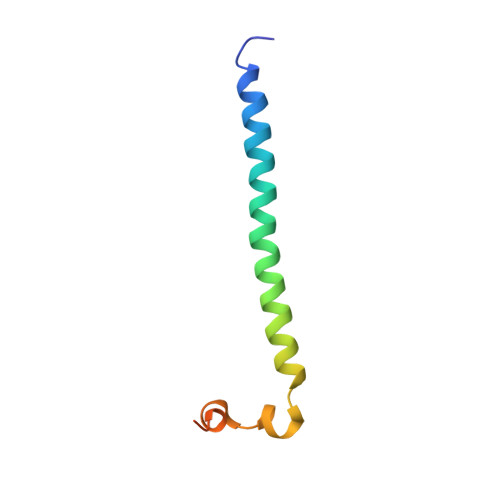
The mitotic regulator Survivin binds as a monomer to its functional interactor Borealin.
Bourhis, E., Hymowitz, S.G., Cochran, A.G.(2007) J Biological Chem 282: 35018-35023
- PubMed: 17881355 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M706233200
- Primary Citation Related Structures:
2RAW, 2RAX - PubMed Abstract:
Survivin is a member of the IAP (inhibitor of apoptosis) protein family, defined in part by the presence of a zinc-binding baculoviral inhibitory repeat (BIR) domain. Most BIR domains bind short sequences beginning with alanine, and in this manner, they recognize and block the action of key targets in apoptotic pathways. However, Survivin binds only very weakly to typical IAP ligands. Unique features of Survivin are the long C-terminal helix following the BIR domain and a short segment (linking the helix and BIR domains) that mediates Survivin homodimerization. Despite this detailed knowledge of the structure of Survivin itself, there is a current lack of understanding about how Survivin recognizes cellular binding partners, and consequently, many questions about Survivin function remain unanswered. We determined two co-crystal structures of Survivin and a minimal binding fragment from the chromosomal passenger protein Borealin, a well validated functional interactor. The interaction between Survivin and Borealin involves extensive packing between the long C-terminal helix of Survivin and a long Borealin helix. Surprisingly, an additional important interaction occurs between the Survivin homodimerization interface and a short segment of Borealin. This segment both structurally mimics and displaces one Survivin monomer. The relevance of this unexpected interaction was tested by mutagenesis of two key Borealin residues. Mutant Borealin introduced into HeLa cells failed to localize properly during mitosis and also caused mislocalization of other chromosomal passenger proteins. This suggests that the mutant is dominant-negative and confirms the functional importance of the interaction surface identified in the crystal structures.
- Department of Protein Engineering, Genentech, Incorporated, South San Francisco, California 94080, USA.
Organizational Affiliation: