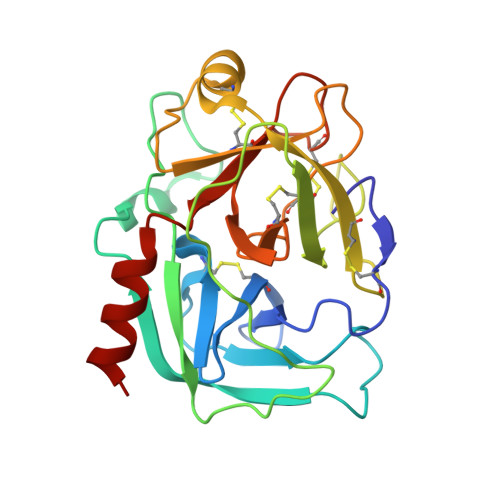
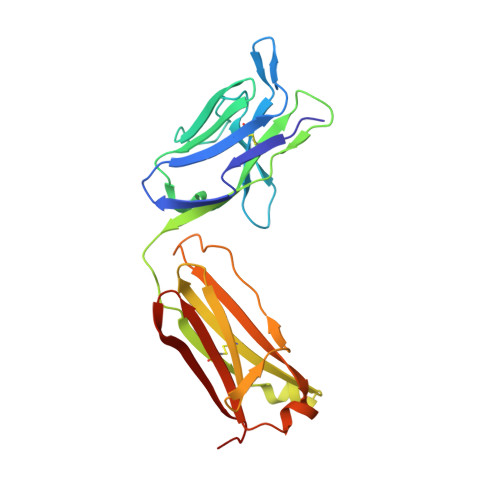
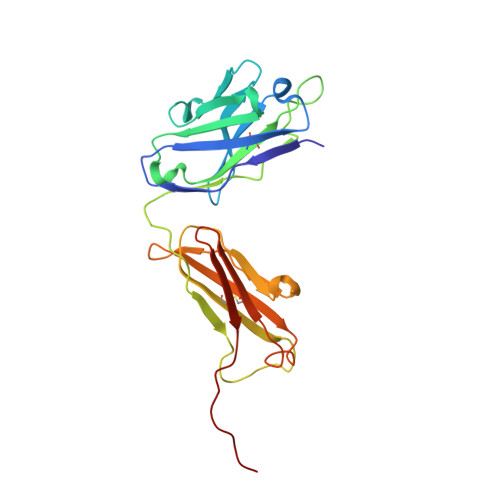
Crystal structure of a ternary complex between human prostate-specific antigen, its substrate acyl intermediate and an activating antibody
Michel, S., Muller, B.H., Bossus, M., Ducancel, F., Jolivet-Reynaud, C., Stura, E.A.(2008) J Mol Biology 376: 1021-1033
- PubMed: 18187150 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2007.11.052
- Primary Citation Related Structures:
2ZCH, 2ZCK, 2ZCL - PubMed Abstract:
Human prostate-specific antigen (PSA or KLK3) is an important marker for the diagnosis and management of prostate cancer. This is an androgen-regulated glycoprotein of the kallikrein-related protease family secreted by prostatic epithelial cells. Its physiological function is to cleave semenogelins in the seminal coagulum and its enzymatic activity is strongly modulated by zinc ions. Here we present the first crystal structure of human PSA in complex with monoclonal antibody (mAb) 8G8F5 that enhances its enzymatic activity. The mAb recognizes an epitope composed of five discontinuous segments including residues from the kallikrein loop and stabilizes PSA in an "open and active conformation" that accelerates catalysis. We also present the crystal structure of PSA in complex with both the mAb 8G8F5 and a fluorogenic substrate Mu-KGISSQY-AFC, derived from semenogelin I. By exploiting the inhibition of PSA by zinc ions, we were able to obtain a substrate acyl intermediate covalently linked to the catalytic serine, at pH 7.3 but not at pH 5.5. Moreover, the inhibition of PSA activity by zinc was found to be modulated by pH variations but not by the antibody binding. The correlation of the different data with the physiological conditions under which PSA can cleave semenogelins is discussed.
- CEA, iBiTec-S/SIMOPRO, LTMB, Gif-sur-Yvette, F-91191, France.
Organizational Affiliation: