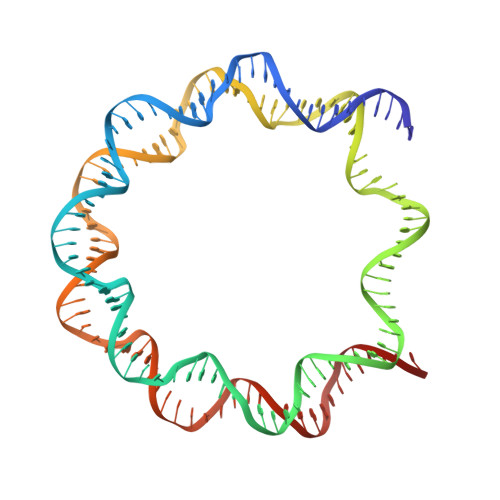
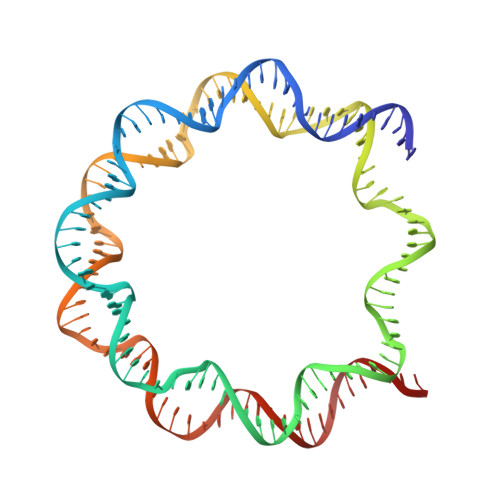
The mechanics behind DNA sequence-dependent properties of the nucleosome
Chua, E.Y.D., Vasudevan, D., Davey, G.E., Wu, B., Davey, C.A.(2012) Nucleic Acids Res 40: 6338-6352
- PubMed: 22453276
- DOI: https://doi.org/10.1093/nar/gks261
- Primary Citation Related Structures:
3UT9, 3UTA, 3UTB - PubMed Abstract:
Chromatin organization and composition impart sophisticated regulatory features critical to eukaryotic genomic function. Although DNA sequence-dependent histone octamer binding is important for nucleosome activity, many aspects of this phenomenon have remained elusive. We studied nucleosome structure and stability with diverse DNA sequences, including Widom 601 derivatives with the highest known octamer affinities, to establish a simple model behind the mechanics of sequence dependency. This uncovers the unique but unexpected role of TA dinucleotides and a propensity for G|C-rich sequence elements to conform energetically favourably at most locations around the histone octamer, which rationalizes G|C% as the most predictive factor for nucleosome occupancy in vivo. In addition, our findings reveal dominant constraints on double helix conformation by H3-H4 relative to H2A-H2B binding and DNA sequence context-dependency underlying nucleosome structure, positioning and stability. This provides a basis for improved prediction of nucleosomal properties and the design of tailored DNA constructs for chromatin investigations.
- Division of Structural and Computational Biology, School of Biological Sciences, Nanyang Technological University, 60 Nanyang Drive, Singapore 637551, Singapore.
Organizational Affiliation: