Structures of bacterial homologues of SWEET transporters in two distinct conformations.
Xu, Y., Tao, Y., Cheung, L.S., Fan, C., Chen, L.Q., Xu, S., Perry, K., Frommer, W.B., Feng, L.(2014) Nature 515: 448-452
- PubMed: 25186729
- DOI: https://doi.org/10.1038/nature13670
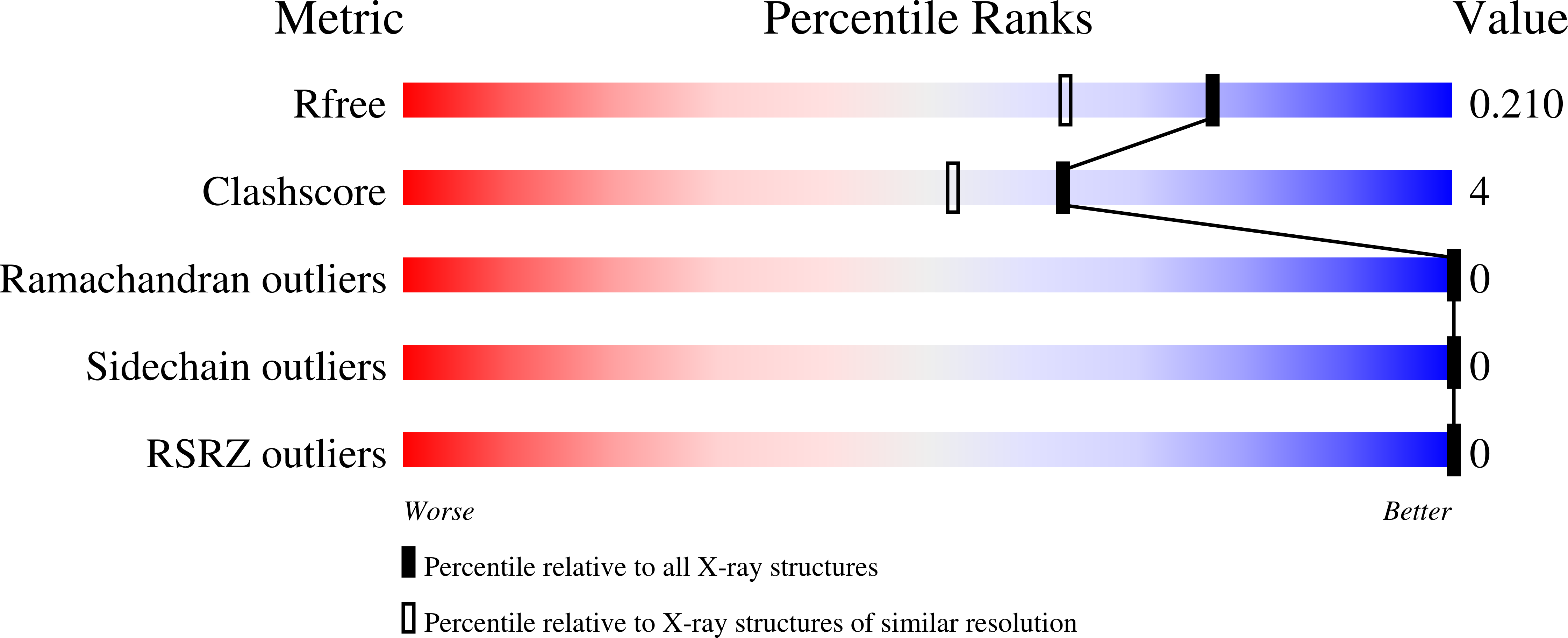
- Primary Citation of Related Structures:
4QNC, 4QND - PubMed Abstract:
SWEETs and their prokaryotic homologues are monosaccharide and disaccharide transporters that are present from Archaea to plants and humans. SWEETs play crucial roles in cellular sugar efflux processes: that is, in phloem loading, pollen nutrition and nectar secretion. Their bacterial homologues, which are called SemiSWEETs, are among the smallest known transporters. Here we show that SemiSWEET molecules, which consist of a triple-helix bundle, form symmetrical, parallel dimers, thereby generating the translocation pathway. Two SemiSWEET isoforms were crystallized, one in an apparently open state and one in an occluded state, indicating that SemiSWEETs and SWEETs are transporters that undergo rocking-type movements during the transport cycle. The topology of the triple-helix bundle is similar yet distinct to that of the basic building block of animal and plant major facilitator superfamily (MFS) transporters (for example, GLUTs and SUTs). This finding indicates two possibilities: that SWEETs and MFS transporters evolved from an ancestral triple-helix bundle or that the triple-helix bundle represents convergent evolution. In SemiSWEETs and SWEETs, two triple-helix bundles are arranged in a parallel configuration to produce the 6- and 6 + 1-transmembrane-helix pores, respectively. In the 12-transmembrane-helix MFS transporters, four triple-helix bundles are arranged into an alternating antiparallel configuration, resulting in a much larger 2 × 2 triple-helix bundle forming the pore. Given the similarity of SemiSWEETs and SWEETs to PQ-loop amino acid transporters and to mitochondrial pyruvate carriers (MPCs), the structures characterized here may also be relevant to other transporters in the MtN3 clan. The insight gained from the structures of these transporters and from the analysis of mutations of conserved residues will improve the understanding of the transport mechanism, as well as allow comparative studies of the different superfamilies involved in sugar transport and the evolution of transporters in general.
Organizational Affiliation:
Department of Molecular and Cellular Physiology, 279 Campus Drive, Stanford University School of Medicine, Stanford CA 94305.