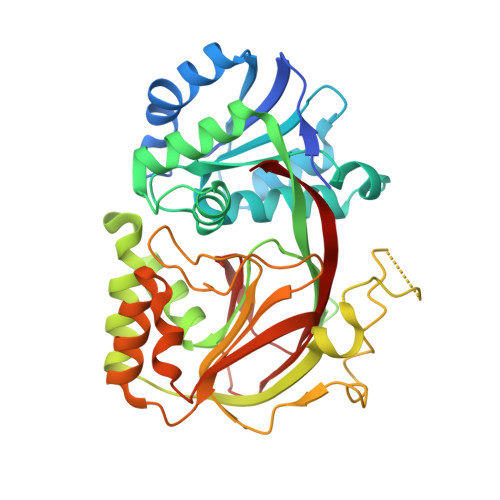
Crystal Structure of Sphingosine Kinase 1 with Pf-543.
Wang, J., Knapp, S., Pyne, N.J., Pyne, S., Elkins, J.M.(2014) ACS Med Chem Lett 5: 1329
- PubMed: 25516793 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/ml5004074
- Primary Citation Related Structures:
4V24 - PubMed Abstract:
The most potent inhibitor of Sphingosine Kinase 1 (SPHK1) so far identified is PF-543. The crystal structure of SPHK1 in complex with inhibitor PF-543 to 1.8 Å resolution reveals the inhibitor bound in a bent conformation analogous to that expected of a bound sphingosine substrate but with a rotated head group. The structural data presented will aid in the design of SPHK1 and SPHK2 inhibitors with improved properties.
- Structural Genomics Consortium, University of Oxford , Old Road Campus Research Building, Old Road Campus, Roosevelt Drive, Oxford OX3 7DQ, U.K.
Organizational Affiliation: