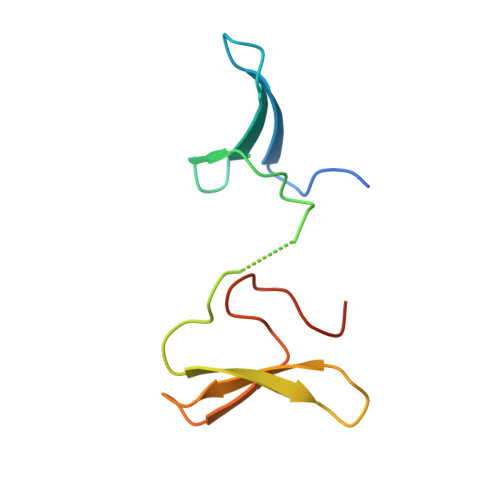
Structural basis for the regulatory role of the PPxY motifs in the thioredoxin-interacting protein TXNIP.
Liu, Y., Lau, J., Li, W., Tempel, W., Li, L., Dong, A., Narula, A., Qin, S., Min, J.(2016) Biochem J 473: 179-187
- PubMed: 26527736 Search on PubMed
- DOI: https://doi.org/10.1042/BJ20150830
- Primary Citation Related Structures:
5CQ2, 5DF6 - PubMed Abstract:
TXNIP (thioredoxin-interacting protein) negatively regulates the antioxidative activity of thioredoxin and participates in pleiotropic cellular processes. Its deregulation is linked to various human diseases, including diabetes, acute myeloid leukaemia and cardiovascular diseases. The E3 ubiquitin ligase Itch (Itchy homologue) polyubiquitinates TXNIP to promote its degradation via the ubiquitin-proteasome pathway, and this Itch-mediated polyubiquitination of TXNIP is dependent on the interaction of the four WW domains of Itch with the two PPxY motifs of TXNIP. However, the molecular mechanism of this interaction of TXNIP with Itch remains elusive. In the present study, we found that each of the four WW domains of Itch exhibited different binding affinities for TXNIP, whereas multivalent engagement between the four WW domains of Itch and the two PPxY motifs of TXNIP resulted in their strong binding avidity. Our structural analyses demonstrated that the third and fourth WW domains of Itch were able to recognize both PPxY motifs of TXNIP simultaneously, supporting a multivalent binding mode between Itch and TXNIP. Interestingly, the phosphorylation status on the tyrosine residue of the PPxY motifs of TXNIP serves as a molecular switch in its choice of binding partners and thereby downstream biological signalling outcomes. Phosphorylation of this tyrosine residue of TXNIP diminished the binding capability of PPxY motifs of TXNIP to Itch, whereas this phosphorylation is a prerequisite to the binding activity of TXNIP to SHP2 [SH2 (Src homology 2) domain-containing protein tyrosine phosphatase 2] and their roles in stabilizing the phosphorylation and activation of CSK (c-Src tyrosine kinase).
- Hubei Key Laboratory of Genetic Regulation and Integrative Biology, College of Life Science, Central China Normal University, Wuhan 430079, P.R. China Structural Genomics Consortium, University of Toronto, 101 College Street, Toronto, Ontario, Canada, M5G 1L7.
Organizational Affiliation: