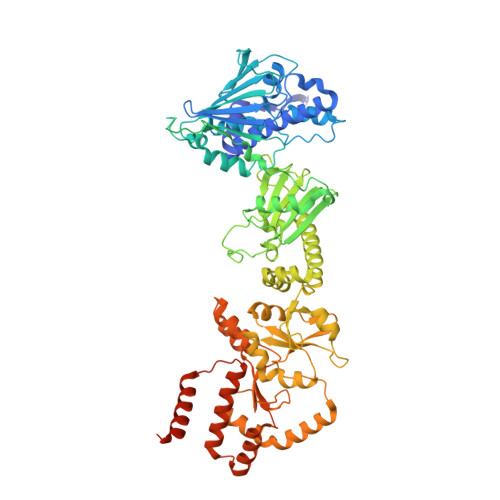
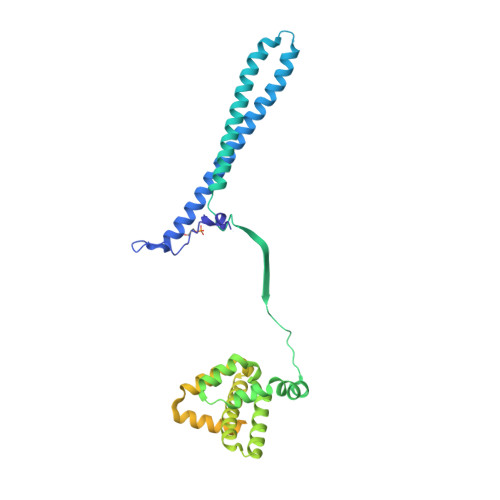
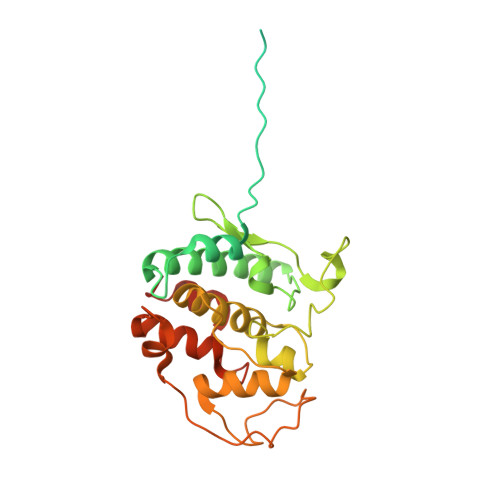
Atomic Structure of Hsp90:Cdc37:Cdk4 Reveals that Hsp90 Traps and Stabilizes an Unfolded Kinase.
Verba, K.A., Wang, R.Y.R., Arakawa, A., Liu, Y., Yokoyama, S., Agard, D.A.(2016) Science 352: 1542
- PubMed: 27339980 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/science.aaf5023
- Primary Citation Related Structures:
5FWK, 5FWL, 5FWM, 5FWP - PubMed Abstract:
The Hsp90 molecular chaperone and its Cdc37 cochaperone help stabilize and activate more than half of the human kinome. However, both the mechanism by which these chaperones assist their "client" kinases and the reason why some kinases are addicted to Hsp90 while closely related family members are independent are unknown. Our structural understanding of these interactions is lacking, as no full-length structures of human Hsp90, Cdc37, or either of these proteins with a kinase have been elucidated. Here we report a 3.9 angstrom cryo-electron microscopy structure of the Hsp90-Cdc37-Cdk4 kinase complex. Surprisingly, the two lobes of Cdk4 are completely separated with the β4-β5 sheet unfolded. Cdc37 mimics part of the kinase N lobe, stabilizing an open kinase conformation by wedging itself between the two lobes. Finally, Hsp90 clamps around the unfolded kinase β5 strand and interacts with exposed N- and C-lobe interfaces, protecting the kinase in a trapped unfolded state. On the basis of this structure and an extensive amount of previously collected data, we propose unifying conceptual and mechanistic models of chaperone-kinase interactions.
- Howard Hughes Medical Institute (HHMI) and the Department of Biochemistry and Biophysics, University of California San Francisco, San Francisco, CA 94158, USA.
Organizational Affiliation: