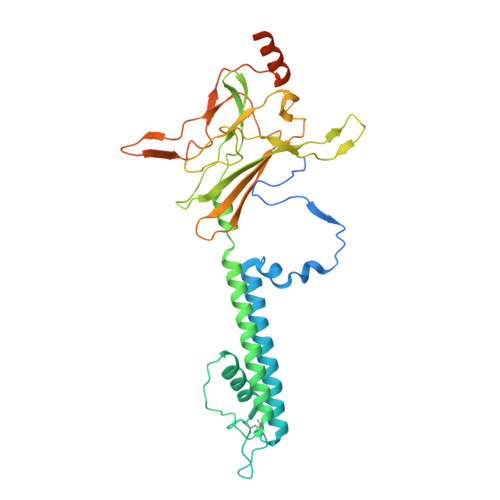
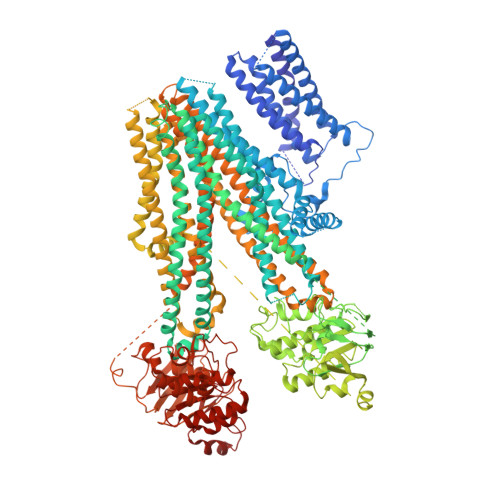
Ligand binding and conformational changes of SUR1 subunit in pancreatic ATP-sensitive potassium channels.
Wu, J.X., Ding, D., Wang, M., Kang, Y., Zeng, X., Chen, L.(2018) Protein Cell 9: 553-567
- PubMed: 29594720 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1007/s13238-018-0530-y
- Primary Citation Related Structures:
5YKE, 5YKF, 5YKG, 5YW7, 5YW8, 5YW9, 5YWA, 5YWB, 5YWC, 5YWD - PubMed Abstract:
ATP-sensitive potassium channels (K ATP ) are energy sensors on the plasma membrane. By sensing the intracellular ADP/ATP ratio of β-cells, pancreatic K ATP channels control insulin release and regulate metabolism at the whole body level. They are implicated in many metabolic disorders and diseases and are therefore important drug targets. Here, we present three structures of pancreatic K ATP channels solved by cryo-electron microscopy (cryo-EM), at resolutions ranging from 4.1 to 4.5 Å. These structures depict the binding site of the antidiabetic drug glibenclamide, indicate how Kir6.2 (inward-rectifying potassium channel 6.2) N-terminus participates in the coupling between the peripheral SUR1 (sulfonylurea receptor 1) subunit and the central Kir6.2 channel, reveal the binding mode of activating nucleotides, and suggest the mechanism of how Mg-ADP binding on nucleotide binding domains (NBDs) drives a conformational change of the SUR1 subunit.
- State Key Laboratory of Membrane Biology, Institute of Molecular Medicine, Peking University, Beijing Key Laboratory of Cardiometabolic Molecular Medicine, Beijing, 100871, China.
Organizational Affiliation: