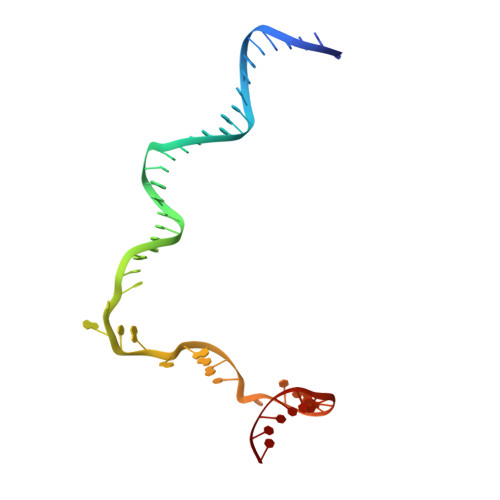
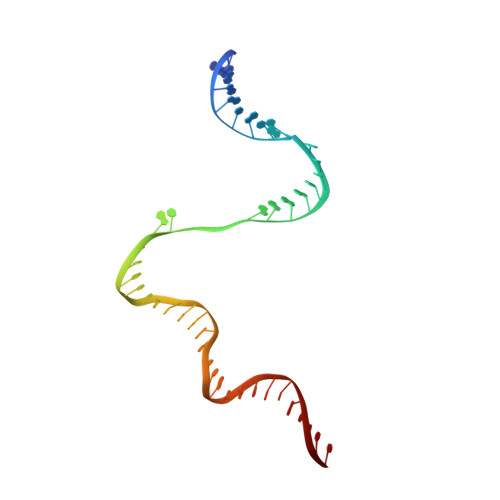
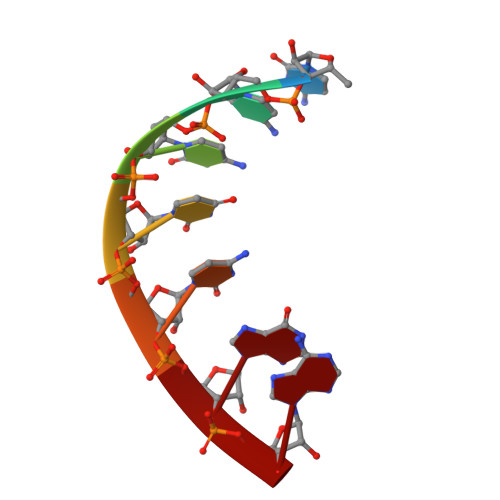
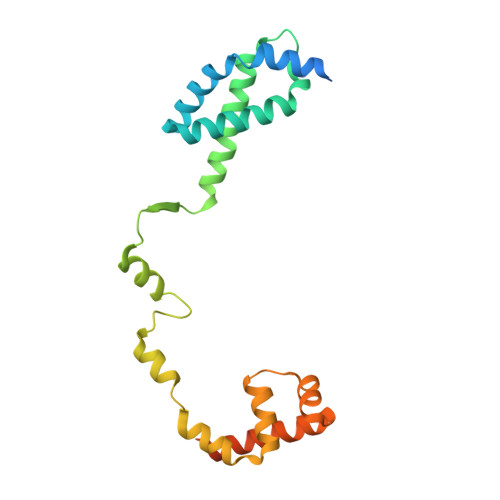Structural basis for transcription initiation by bacterial ECF sigma factors.
Li, L., Fang, C., Zhuang, N., Wang, T., Zhang, Y.(2019) Nat Commun 10: 1153-1153
- PubMed: 30858373 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-019-09096-y
- Primary Citation Related Structures:
5ZX2, 5ZX3 - PubMed Abstract:
Bacterial RNA polymerase employs extra-cytoplasmic function (ECF) σ factors to regulate context-specific gene expression programs. Despite being the most abundant and divergent σ factor class, the structural basis of ECF σ factor-mediated transcription initiation remains unknown. Here, we determine a crystal structure of Mycobacterium tuberculosis (Mtb) RNAP holoenzyme comprising an RNAP core enzyme and the ECF σ factor σ H (σ H -RNAP) at 2.7 Å, and solve another crystal structure of a transcription initiation complex of Mtb σ H -RNAP (σ H -RPo) comprising promoter DNA and an RNA primer at 2.8 Å. The two structures together reveal the interactions between σ H and RNAP that are essential for σ H -RNAP holoenzyme assembly as well as the interactions between σ H -RNAP and promoter DNA responsible for stringent promoter recognition and for promoter unwinding. Our study establishes that ECF σ factors and primary σ factors employ distinct mechanisms for promoter recognition and for promoter unwinding.
- Key Laboratory of Synthetic Biology, CAS Center for Excellence in Molecular Plant Sciences, Shanghai Institute of Plant Physiology and Ecology, Chinese Academy of Sciences, Shanghai, 200032, China.
Organizational Affiliation: