The molybdenum storage protein - A bionanolab for creating experimentally alterable polyoxomolybdate clusters.
Brunle, S., Poppe, J., Hail, R., Demmer, U., Ermler, U.(2018) J Inorg Biochem 189: 172-179
Experimental Data Snapshot
Starting Model: experimental
View more details
(2018) J Inorg Biochem 189: 172-179
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Molybdenum storage protein subunit beta | A [auth B] | 269 | Azotobacter vinelandii DJ | Mutation(s): 0 Gene Names: mosB, Avin_43210 | 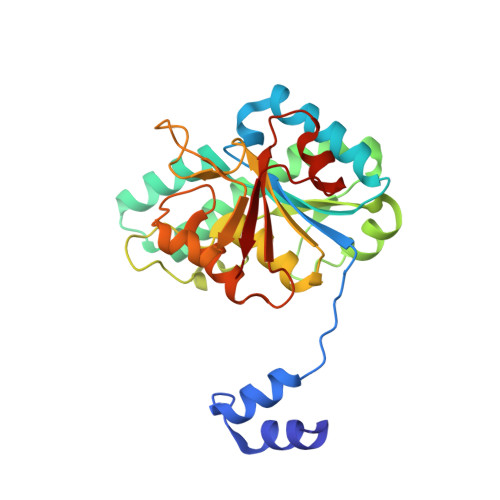 |
UniProt | |||||
Find proteins for P84253 (Azotobacter vinelandii (strain DJ / ATCC BAA-1303)) Explore P84253 Go to UniProtKB: P84253 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P84253 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Molybdenum storage protein subunit alpha | B [auth A] | 275 | Azotobacter vinelandii DJ | Mutation(s): 0 Gene Names: mosA, Avin_43200 | 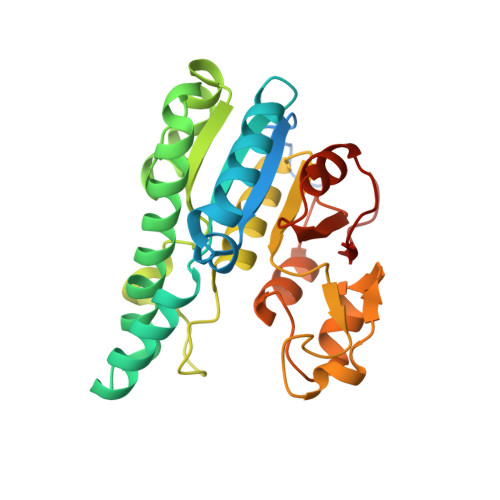 |
UniProt | |||||
Find proteins for P84308 (Azotobacter vinelandii (strain DJ / ATCC BAA-1303)) Explore P84308 Go to UniProtKB: P84308 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P84308 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ATP Query on ATP | F [auth A] | ADENOSINE-5'-TRIPHOSPHATE C10 H16 N5 O13 P3 ZKHQWZAMYRWXGA-KQYNXXCUSA-N |  | ||
| PO4 Query on PO4 | C [auth B] D [auth B] H [auth A] I [auth A] J [auth A] | PHOSPHATE ION O4 P NBIIXXVUZAFLBC-UHFFFAOYSA-K |  | ||
| CL Query on CL | E [auth B] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| MG Query on MG | G [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 115.4 | α = 90 |
| b = 115.4 | β = 90 |
| c = 234.5 | γ = 120 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| XSCALE | data scaling |
| PHENIX | phasing |