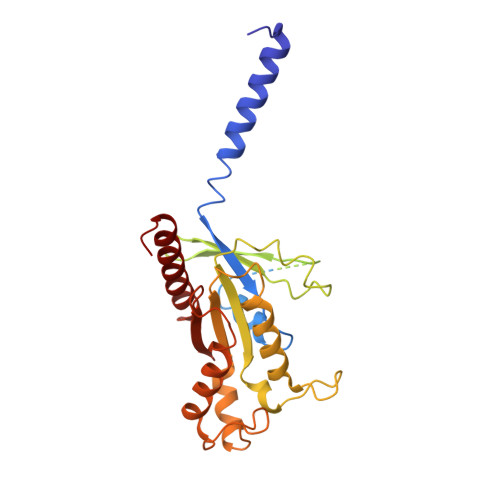
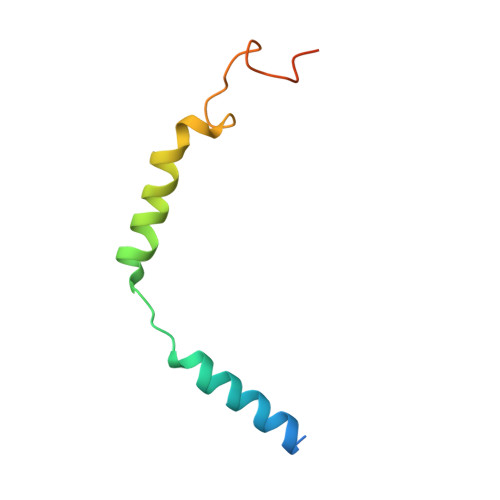
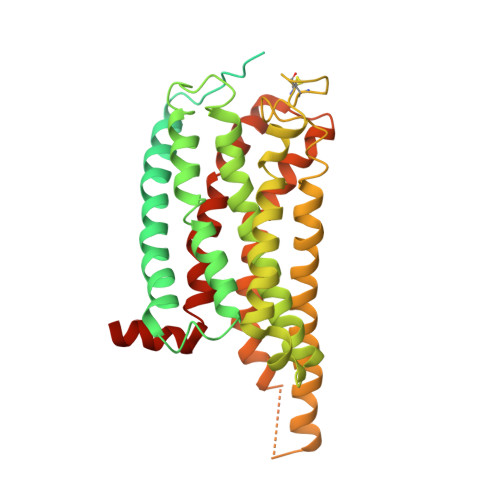
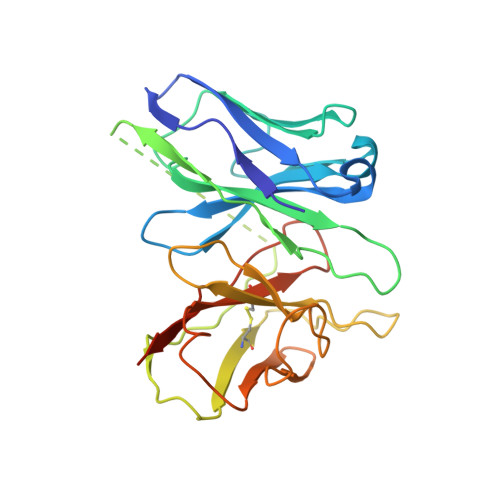
Activation and Signaling Mechanism Revealed by Cannabinoid Receptor-GiComplex Structures.
Hua, T., Li, X., Wu, L., Iliopoulos-Tsoutsouvas, C., Wang, Y., Wu, M., Shen, L., Johnston, C.A., Nikas, S.P., Song, F., Song, X., Yuan, S., Sun, Q., Wu, Y., Jiang, S., Grim, T.W., Benchama, O., Stahl, E.L., Zvonok, N., Zhao, S., Bohn, L.M., Makriyannis, A., Liu, Z.J.(2020) Cell 180: 655
- PubMed: 32004463 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.cell.2020.01.008
- Primary Citation Related Structures:
6KPC, 6KPF, 6KPG - PubMed Abstract:
Human endocannabinoid systems modulate multiple physiological processes mainly through the activation of cannabinoid receptors CB1 and CB2. Their high sequence similarity, low agonist selectivity, and lack of activation and G protein-coupling knowledge have hindered the development of therapeutic applications. Importantly, missing structural information has significantly held back the development of promising CB2-selective agonist drugs for treating inflammatory and neuropathic pain without the psychoactivity of CB1. Here, we report the cryoelectron microscopy structures of synthetic cannabinoid-bound CB2 and CB1 in complex with G i , as well as agonist-bound CB2 crystal structure. Of important scientific and therapeutic benefit, our results reveal a diverse activation and signaling mechanism, the structural basis of CB2-selective agonists design, and the unexpected interaction of cholesterol with CB1, suggestive of its endogenous allosteric modulating role.
- iHuman Institute, ShanghaiTech University, Shanghai 201210, China. Electronic address: huatian@shanghaitech.edu.cn.
Organizational Affiliation: