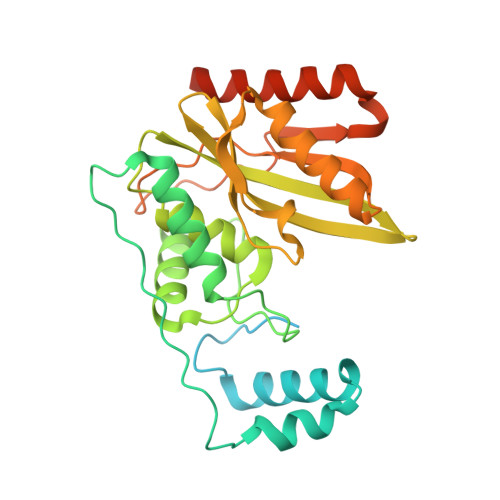
Crystal structure of the tubulin tyrosine carboxypeptidase complex VASH1-SVBP.
Adamopoulos, A., Landskron, L., Heidebrecht, T., Tsakou, F., Bleijerveld, O.B., Altelaar, M., Nieuwenhuis, J., Celie, P.H.N., Brummelkamp, T.R., Perrakis, A.(2019) Nat Struct Mol Biol 26: 567-570
- PubMed: 31270470 Search on PubMed
- DOI: https://doi.org/10.1038/s41594-019-0254-6
- Primary Citation Related Structures:
6NVQ - PubMed Abstract:
The cyclic enzymatic removal and ligation of the C-terminal tyrosine of α-tubulin generates heterogeneous microtubules and affects their functions. Here we describe the crystal and solution structure of the tubulin carboxypeptidase complex between vasohibin (VASH1) and small vasohibin-binding protein (SVBP), which folds in a long helix, which stabilizes the VASH1 catalytic domain. This structure, combined with molecular docking and mutagenesis experiments, reveals which residues are responsible for recognition and cleavage of the tubulin C-terminal tyrosine.
- Division of Biochemistry, the Netherlands Cancer Institute, Amsterdam, the Netherlands.
Organizational Affiliation: