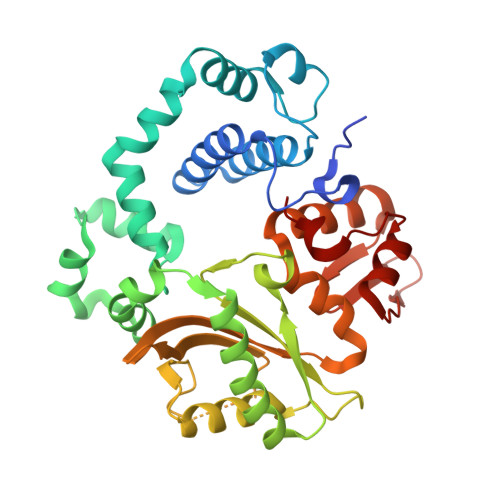
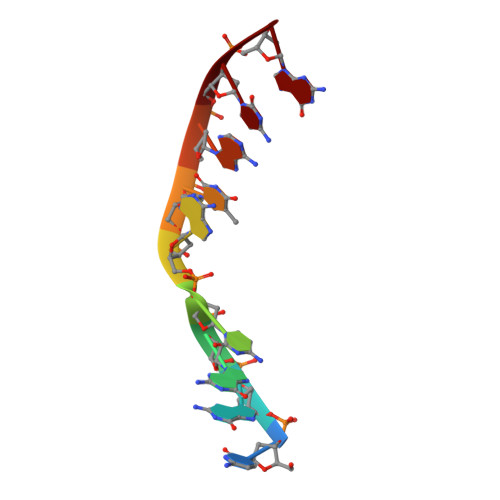
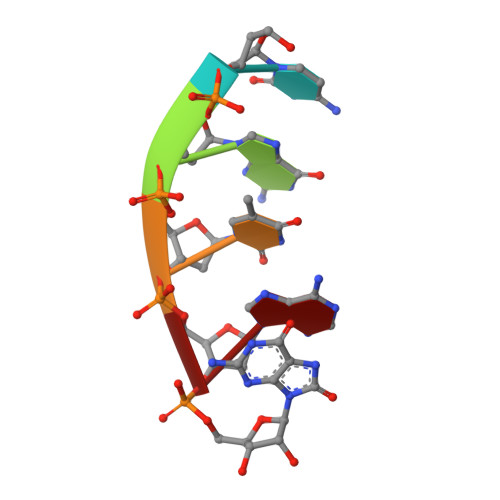
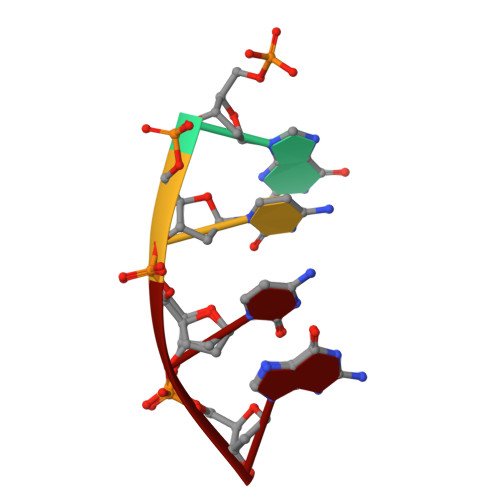
Structural basis for proficient oxidized ribonucleotide insertion in double strand break repair.
Jamsen, J.A., Sassa, A., Perera, L., Shock, D.D., Beard, W.A., Wilson, S.H.(2021) Nat Commun 12: 5055-5055
- PubMed: 34417448 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-021-24486-x
- Primary Citation Related Structures:
6VEZ, 6VF0, 6VF1, 6VF2, 6VF3, 6VF4, 6VF5, 6VF6, 6VF7, 6VF8, 6VF9, 6VFA, 6VFB, 6VFC - PubMed Abstract:
Reactive oxygen species (ROS) oxidize cellular nucleotide pools and cause double strand breaks (DSBs). Non-homologous end-joining (NHEJ) attaches broken chromosomal ends together in mammalian cells. Ribonucleotide insertion by DNA polymerase (pol) μ prepares breaks for end-joining and this is required for successful NHEJ in vivo. We previously showed that pol μ lacks discrimination against oxidized dGTP (8-oxo-dGTP), that can lead to mutagenesis, cancer, aging and human disease. Here we reveal the structural basis for proficient oxidized ribonucleotide (8-oxo-rGTP) incorporation during DSB repair by pol μ. Time-lapse crystallography snapshots of structural intermediates during nucleotide insertion along with computational simulations reveal substrate, metal and side chain dynamics, that allow oxidized ribonucleotides to escape polymerase discrimination checkpoints. Abundant nucleotide pools, combined with inefficient sanitization and repair, implicate pol μ mediated oxidized ribonucleotide insertion as an emerging source of widespread persistent mutagenesis and genomic instability.
- Genome Integrity and Structural Biology Laboratory, National Institute of Environmental Health Sciences, National Institutes of Health, Research Triangle Park, NC, USA. joonas.jamsen@nih.gov.
Organizational Affiliation: