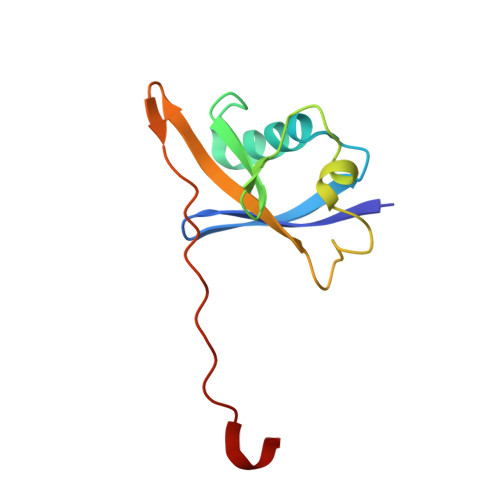
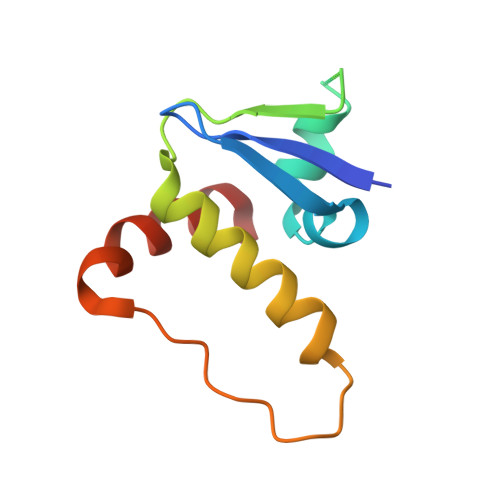
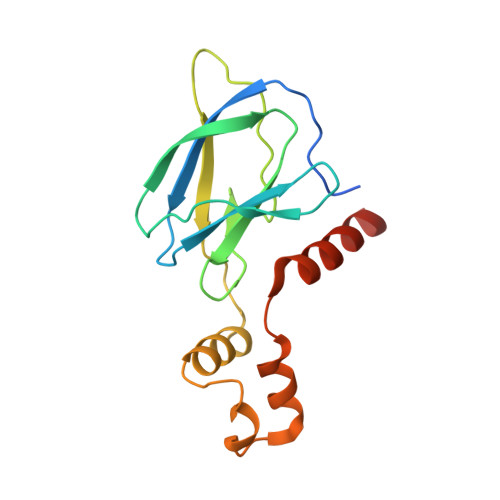
Tracking the PROTAC degradation pathway in living cells highlights the importance of ternary complex measurement for PROTAC optimization.
Schwalm, M.P., Kramer, A., Dolle, A., Weckesser, J., Yu, X., Jin, J., Saxena, K., Knapp, S.(2023) Cell Chem Biol 30: 753-765.e8
- PubMed: 37354907 Search on PubMed
- DOI: https://doi.org/10.1016/j.chembiol.2023.06.002
- Primary Citation Related Structures:
7Q2J, 8BB4, 8BB5, 8C13 - PubMed Abstract:
The multi-step degradation process of PROteolysis TArgeting Chimeras (PROTACs) poses a challenge for their rational development, as the rate-limiting steps that determine PROTACs efficiency remain largely unknown. Moreover, the slow throughput of currently used endpoint assays does not allow the comprehensive analysis of larger series of PROTACs. Here, we developed cell-based assays using the NanoLuciferase and HaloTag that allow measuring PROTAC-induced degradation and ternary complex formation kinetics and stability in cells. Using PROTACs developed for the degradation of WD40 repeat domain protein 5 (WDR5), the characterization of the mode of action of these PROTACs in the early degradation cascade revealed a key role of ternary complex formation and stability. Comparing a series of ternary complex crystal structures highlighted the importance of an efficient E3-target interface for ternary complex stability. The developed assays outline a strategy for the rational optimization of PROTACs using a series of live cell assays monitoring key steps of the early PROTAC-induced degradation pathway.
- Institut für Pharmazeutische Chemie, Goethe-University Frankfurt, Biozentrum, Max-von-Laue-Str. 9, 60438 Frankfurt am Main, Germany; Structural Genomics Consortium, Goethe-University Frankfurt, Buchmann Institute for Life Sciences, Max-von-Laue-Str. 15, 60438 Frankfurt am Main, Germany.
Organizational Affiliation: