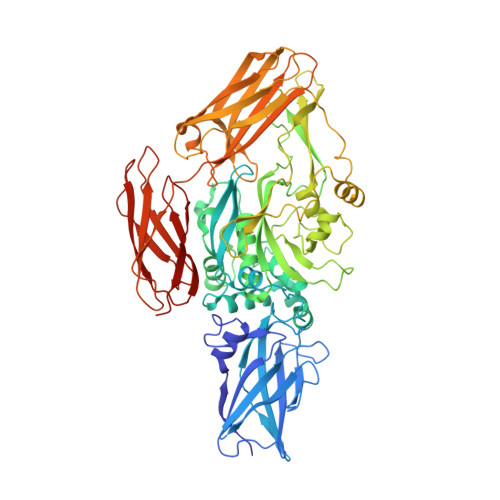
Distinct conformational states enable transglutaminase 2 to promote cancer cell survival versus cell death.
Aplin, C., Zielinski, K.A., Pabit, S., Ogunribido, D., Katt, W.P., Pollack, L., Cerione, R.A., Milano, S.K.(2024) Commun Biol 7: 982-982
- PubMed: 39134806 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s42003-024-06672-x
- Primary Citation Related Structures:
8TR9 - PubMed Abstract:
Transglutaminase 2 (TG2) is a GTP-binding, protein-crosslinking enzyme that has been investigated as a therapeutic target for Celiac disease, neurological disorders, and aggressive cancers. TG2 has been suggested to adopt two conformational states that regulate its functions: a GTP-bound, closed conformation, and a calcium-bound, crosslinking-active open conformation. TG2 mutants that constitutively adopt an open conformation are cytotoxic to cancer cells. Thus, small molecules that bind and stabilize the open conformation of TG2 could offer a new therapeutic strategy. Here, we investigate TG2, using static and time-resolved small-angle X-ray scattering (SAXS) and single-particle cryoelectron microscopy (cryo-EM), to determine the conformational states responsible for conferring its biological effects. We also describe a newly developed TG2 inhibitor, LM11, that potently kills glioblastoma cells and use SAXS to investigate how LM11 affects the conformational states of TG2. Using SAXS and cryo-EM, we show that guanine nucleotides bind and stabilize a monomeric closed conformation while calcium binds to an open state that can form higher order oligomers. SAXS analysis suggests how a TG2 mutant that constitutively adopts the open state binds nucleotides through an alternative mechanism to wildtype TG2. Furthermore, we use time resolved SAXS to show that LM11 increases the ability of calcium to bind and stabilize an open conformation, which is not reversible by guanine nucleotides and is cytotoxic to cancer cells. Taken together, our findings demonstrate that the conformational dynamics of TG2 are more complex than previously suggested and highlight how conformational stabilization of TG2 by LM11 maintains TG2 in a cytotoxic conformational state.
- Department of Chemistry and Chemical Biology, Cornell University, 14853, Ithaca, NY, USA.
Organizational Affiliation: