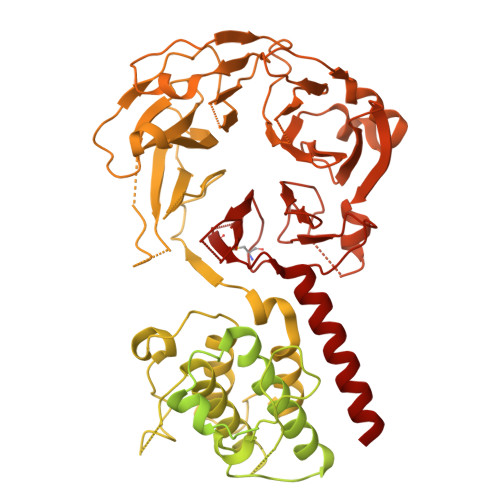
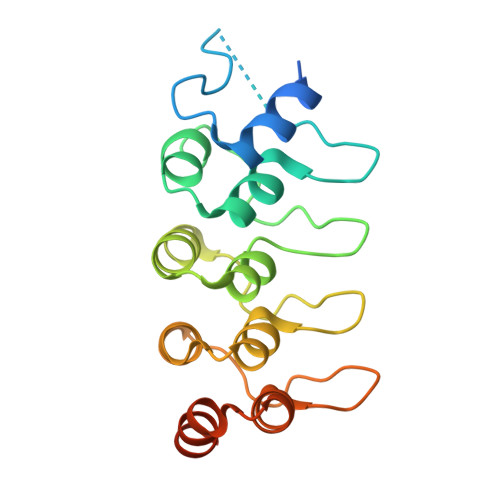
A designed ankyrin-repeat protein that targets Parkinson's disease-associated LRRK2.
Dederer, V., Sanz Murillo, M., Karasmanis, E.P., Hatch, K.S., Chatterjee, D., Preuss, F., Abdul Azeez, K.R., Nguyen, L.V., Galicia, C., Dreier, B., Pluckthun, A., Versees, W., Mathea, S., Leschziner, A.E., Reck-Peterson, S.L., Knapp, S.(2024) J Biological Chem 300: 107469-107469
- PubMed: 38876305
- DOI: https://doi.org/10.1016/j.jbc.2024.107469
- Primary Citation of Related Structures:
8U1B - PubMed Abstract:
Leucine rich repeat kinase 2 (LRRK2) is a large multidomain protein containing two catalytic domains, a kinase and a GTPase, as well as protein interactions domains, including a WD40 domain. The association of increased LRRK2 kinase activity with both the familial and sporadic forms of Parkinson's disease has led to an intense interest in determining its cellular function. However, small molecule probes that can bind to LRRK2 and report on or affect its cellular activity are needed. Here, we report the identification and characterization of the first high-affinity LRRK2-binding designed ankyrin-repeat protein (DARPin), named E11. Using cryo-EM, we show that DARPin E11 binds to the LRRK2 WD40 domain. LRRK2 bound to DARPin E11 showed improved behavior on cryo-EM grids, resulting in higher resolution LRRK2 structures. DARPin E11 did not affect the catalytic activity of a truncated form of LRRK2 in vitro but decreased the phosphorylation of Rab8A, a LRRK2 substrate, in cells. We also found that DARPin E11 disrupts the formation of microtubule-associated LRRK2 filaments in cells, which are known to require WD40-based dimerization. Thus, DARPin E11 is a new tool to explore the function and dysfunction of LRRK2 and guide the development of LRRK2 kinase inhibitors that target the WD40 domain instead of the kinase.
- Institute of Pharmaceutical Chemistry, Goethe-Universität, Frankfurt, Germany; Structural Genomics Consortium (SGC), Buchmann Institute for Life Sciences, Goethe-Universität, Frankfurt, Germany; Aligning Science Across Parkinson's (ASAP), Chevy Chase, Maryland, USA.
Organizational Affiliation: