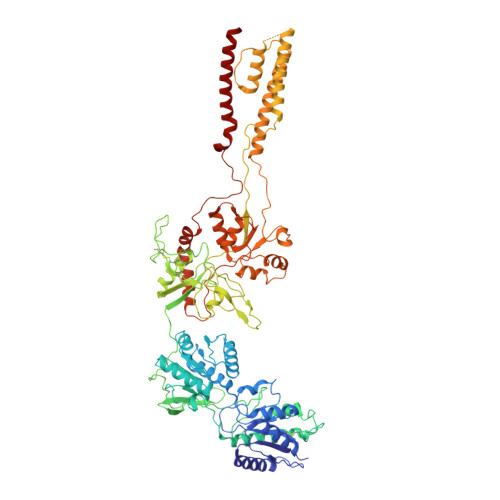
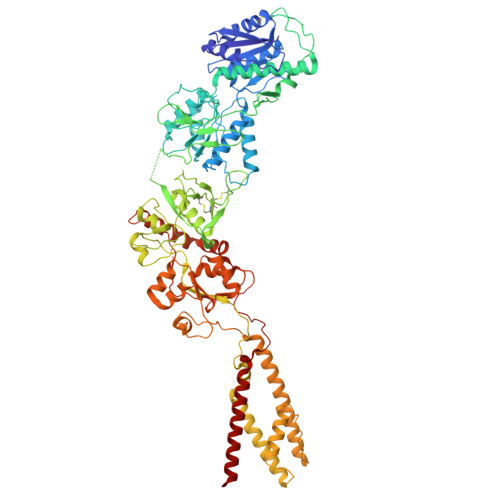
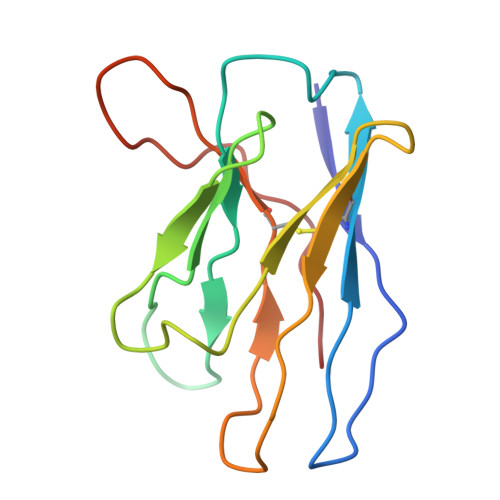
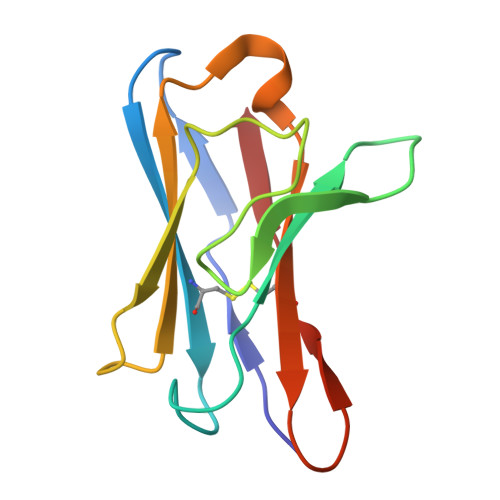
Structural and functional mechanisms of anti-NMDAR autoimmune encephalitis.
Michalski, K., Abdulla, T., Kleeman, S., Schmidl, L., Gomez, R., Simorowski, N., Vallese, F., Pruss, H., Heckmann, M., Geis, C., Furukawa, H.(2024) Nat Struct Mol Biol 31: 1975-1986
- PubMed: 39227719 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-024-01386-4
- Primary Citation Related Structures:
8VUH, 8VUJ, 8VUL, 8VUN, 8VUQ, 8VUR, 8VUS, 8VUT, 8VUU, 8VUV, 8VUY, 8VVH - PubMed Abstract:
Autoantibodies against neuronal membrane proteins can manifest in autoimmune encephalitis, inducing seizures, cognitive dysfunction and psychosis. Anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis is the most dominant autoimmune encephalitis; however, insights into how autoantibodies recognize and alter receptor functions remain limited. Here we determined structures of human and rat NMDARs bound to three distinct patient-derived antibodies using single-particle electron cryo-microscopy. These antibodies bind different regions within the amino-terminal domain of the GluN1 subunit. Through electrophysiology, we show that all three autoantibodies acutely and directly reduced NMDAR channel functions in primary neurons. Antibodies show different stoichiometry of binding and antibody-receptor complex formation, which in one antibody, 003-102, also results in reduced synaptic localization of NMDARs. These studies demonstrate mechanisms of diverse epitope recognition and direct channel regulation of anti-NMDAR autoantibodies underlying autoimmune encephalitis.
- W.M. Keck Structural Biology Laboratory, Cold Spring Harbor Laboratory, Cold Spring Harbor, NY, USA.
Organizational Affiliation: