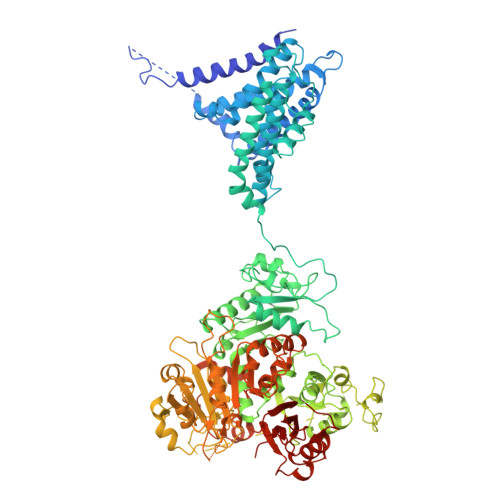
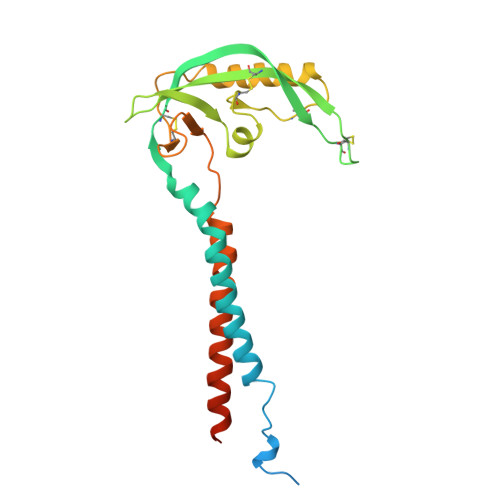
Ball-and-chain inactivation of a human large conductance calcium-activated potassium channel.
Agarwal, S., Kim, E.D., Lee, S., Simon, A., Accardi, A., Nimigean, C.M.(2025) Nat Commun 16: 1769-1769
- PubMed: 39971906 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-56844-4
- Primary Citation Related Structures:
9CZH, 9CZJ, 9CZK, 9CZM, 9CZO, 9CZQ, 9D18, 9D19 - PubMed Abstract:
BK channels are large-conductance calcium (Ca 2+ )-activated potassium channels crucial for neuronal excitability, muscle contraction, and neurotransmitter release. The pore-forming (α) subunits co-assemble with auxiliary (β and γ) subunits that modulate their function. Previous studies demonstrated that the N-termini of β2-subunits can inactivate BK channels, but with no structural correlate. Here, we investigate BK β2-subunit inactivation using cryo-electron microscopy, electrophysiology and molecular dynamics simulations. We find that the β2 N-terminus occludes the pore only in the Ca 2+ -bound open state, via a ball-and-chain mechanism. The first three hydrophobic residues of β2 are crucial for occlusion, while the remainder of the N-terminus remains flexible. Neither the closed channel conformation obtained in the absence of Ca 2+ nor an intermediate conformation found in the presence of Ca 2+ show density for the N-terminus of the β2 subunit in their pore, likely due to narrower side access portals preventing their entry into the channel pore.
- Department of Anesthesiology, Weill Cornell Medical College, 1300 York Ave, New York, NY, USA.
Organizational Affiliation: